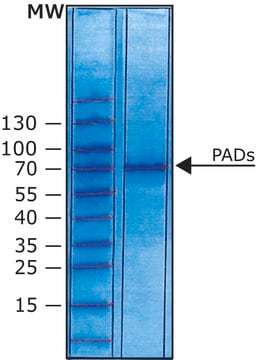
SAE0061
Peptidyl arginine deiminase type-2 (PAD2) human
recombinant, expressed in E. coli
Synonym(e):
PAD-2, PAD2, PADI-2, PADI2
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empfohlene Produkte
Rekombinant
expressed in E. coli
Qualitätsniveau
Spezifische Aktivität
≥40 units/mg protein
Versandbedingung
dry ice
Lagertemp.
−20°C
Verwandte Kategorien
Allgemeine Beschreibung
Peptidyl Arginine Deiminases (PADs) perform post-translational deiminations of proteins. PADs are calcium-dependent enzymes that catalyze the conversion of L-arginine residues to L-citrulline. The reaction catalyzed is as follows:
Protein--[L-arginine] + H2O+Ca+2→Protein--[L-citrulline]+ NH4
This deimination provides another level of regulating protein function.
There are five mammalian PADs sub-types, which differ in substrate specificity and tissue distribution. PAD enzymes are highly homologous, with 50–60% sequence similarity. PADenzymes play important roles in gene regulation by citrullination of arginine residues on histones H3, H2A, and H4. Overexpression of these enzymes has been found in several diseases such as rheumatoid arthritis, Alzheimer′s disease, multiple sclerosis, lupus, Parkinson′s disease, and cancer. PAD2 is widely expressed in the brain, secretory glands, and skeletal muscles. The enzyme was found to be responsible for hyper-citrullination of myelin basic protein, which is suspected to lead to multiple sclerosis. The product is supplied in 20 mM Tris, pH 7.5, 200 mM NaCl, 10% (w/v) glycerol, 10 mM 2-mercaptoethanol, 1 mM EDTA, 1 mM DTT, 10 mM CaCl2, 10 mM arginine, 30 mM maltose, and 0.6 mg/ml dodecyl−β-D-maltoside. Enzymatic activity: Activity is measured by a colorimetric method with the synthetic substrate benzoyl arginine ethyl ester (BAEE).
Protein--[L-arginine] + H2O+Ca+2→Protein--[L-citrulline]+ NH4
This deimination provides another level of regulating protein function.
There are five mammalian PADs sub-types, which differ in substrate specificity and tissue distribution. PAD enzymes are highly homologous, with 50–60% sequence similarity. PADenzymes play important roles in gene regulation by citrullination of arginine residues on histones H3, H2A, and H4. Overexpression of these enzymes has been found in several diseases such as rheumatoid arthritis, Alzheimer′s disease, multiple sclerosis, lupus, Parkinson′s disease, and cancer. PAD2 is widely expressed in the brain, secretory glands, and skeletal muscles. The enzyme was found to be responsible for hyper-citrullination of myelin basic protein, which is suspected to lead to multiple sclerosis. The product is supplied in 20 mM Tris, pH 7.5, 200 mM NaCl, 10% (w/v) glycerol, 10 mM 2-mercaptoethanol, 1 mM EDTA, 1 mM DTT, 10 mM CaCl2, 10 mM arginine, 30 mM maltose, and 0.6 mg/ml dodecyl−β-D-maltoside. Enzymatic activity: Activity is measured by a colorimetric method with the synthetic substrate benzoyl arginine ethyl ester (BAEE).
Einheitendefinition
One unit will produce 1.0 μmole of N-a-benzoylcitrulline ethyl ester from BAEE per hr at 55 °C at pH 7.2.
Physikalische Form
Supplied as a buffered aqueous solution containing Trizma, NaCl, glycerol, mercaptoethanol, EDTA, DTT, CaCl2, arginine, maltose, and dodecyl-β-D-maltoside.
Angaben zur Herstellung
Contains a maltose-binding protein fusion tag.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 1
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Sunish Mohanan et al.
Biochemistry research international, 2012, 895343-895343 (2012-09-29)
The peptidylarginine deiminases (PADs) are a family of posttranslational modification enzymes that catalyze the conversion of positively charged protein-bound arginine and methylarginine residues to the uncharged, nonstandard amino acid citrulline. This enzymatic activity is referred to as citrullination or, alternatively
H Takahara et al.
Journal of biochemistry, 94(6), 1945-1953 (1983-12-01)
The preceding paper described the identification and some properties of peptidylarginine deiminase, which catalyzes the deimination of arginyl residues in protein, from rabbit skeletal muscle, kidney, brain, and lung. In the present work we purified peptidylarginine deiminase from rabbit skeletal
Olena Mahneva et al.
PloS one, 15(1), e0227822-e0227822 (2020-01-16)
Peptidylarginine deiminase (PAD) modifies peptidylarginine and converts it to peptidylcitrulline in the presence of elevated calcium. Protein modification can lead to severe changes in protein structure and function, and aberrant PAD activity is linked to human pathologies. While PAD homologs
Mario A Moscarello et al.
Neurochemical research, 32(2), 251-256 (2006-10-13)
The pathogenesis of MS is unknown. In our studies, we have demonstrated an important role for citrullinated myelin basic protein (MBP). The accompanying loss of positive charge compromises the ability of MBP to interact with the lipid bilayer. The conversion
Kevin L Bicker et al.
Biopolymers, 99(2), 155-163 (2012-11-24)
The post-translational modification of histones has significant effects on overall chromatin function. One such modification is citrullination, which is catalyzed by the protein arginine deiminases (PADs), a unique family of enzymes that catalyzes the hydrolysis of peptidyl-arginine to form peptidyl-citrulline
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.