Alle Fotos(1)
Wichtige Dokumente
M5939
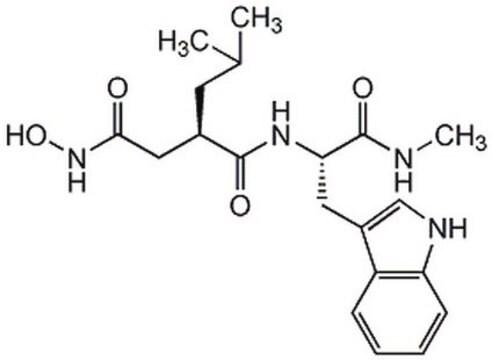
(R)-N4-Hydroxy-N1-[(S)-2-(1H-indol-3-yl)-1-methylcarbamoyl-ethyl]-2-isobutyl-succinamide
>95% (HPLC)
Synonym(e):
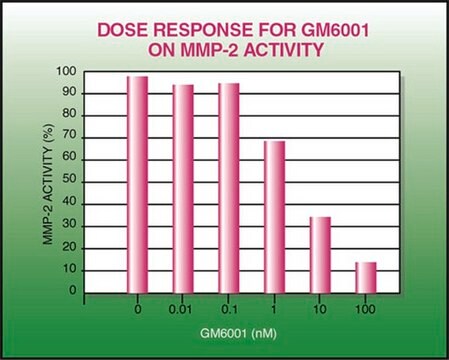
GM6001
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C20H28N4O4
CAS-Nummer:
Molekulargewicht:
388.46
UNSPSC-Code:
12352200
PubChem Substanz-ID:
NACRES:
NA.77
Empfohlene Produkte
Qualitätsniveau
Assay
>95% (HPLC)
Form
solid
Lagertemp.
2-8°C
SMILES String
CNC([C@@H](NC([C@@H](C(C)C)CC(NC)=O)=O)CC1=CNC2=CC=CC=C21)=O
InChI
1S/C20H28N4O3/c1-12(2)15(10-18(25)21-3)19(26)24-17(20(27)22-4)9-13-11-23-16-8-6-5-7-14(13)16/h5-8,11-12,15,17,23H,9-10H2,1-4H3,(H,21,25)(H,22,27)(H,24,26)/t15-,17+/m1/s1
InChIKey
PFBWZAGFLVKAGR-WBVHZDCISA-N
Anwendung
(R)-N4-Hydroxy-N1-[(S)-2-(1H-indol-3-yl)-1-methylcarbamoyl-ethyl]-2-isobutyl-succinamide has been used:
- To study its effect on the profiling of active aggrecanases and their specific aggrecan degradation fragments.
- To study the involvement of autocrine EGF (epidermal growth factor) receptor activation in the regulation of the morphogenetic process, using human umbilical vein endothelial cells.
- To study the effect of GM6001 blockade on the expression of angiotensin II, the interstitial collagenases and soluble elastin fragments in explant culture supernatants.
Biochem./physiol. Wirkung
GM6001 promotes cardiovascular and hepatocellular function.
Inhibits a wide variety of matrix metalloproteases with Kis in the nanomolar range. Active in a number of animal models of diseases where matrix metalloproteases are thought to be involved.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Administration of a matrix metalloproteinase inhibitor after hemorrhage improves cardiovascular and hepatocellular function.
Wang P, et al.
Shock, 6(5), 377-382 (1996)
Role of the angiotensin-converting enzyme 1/angiotensin II/angiotensin receptor 1 axis in interstitial collagenase expression in human carotid atheroma
Clancy P, et al.
Atherosclerosis, 229(2), 331-337 (2013)
Autocrine EGF receptor activation mediates endothelial cell migration and vascular morphogenesis induced by VEGF under interstitial flow
Semino CE, et al.
Experimental Cell Research, 312(3), 289-298 (2006)
The effect of protease inhibitors on the induction of osteoarthritis-related biomarkers in bovine full-depth cartilage explants
He Y, et al.
PLoS ONE, 10(4), e0122700-e0122700 (2015)
D Grobelny et al.
Biochemistry, 31(31), 7152-7154 (1992-08-11)
The hydroxamic acid HONHCOCH2CH(i-Bu)CO-L-Trp-NHMe, isomer 6A (GM 6001), inhibits human skin fibroblast collagenase with Ki of 0.4 nM using the synthetic thiol ester substrate Ac-Pro-Leu-Gly-SCH(i-Bu)CO-Leu-Gly-OEt at pH 6.5. The other isomer, 6B, which has the opposite configuration at the CH2CH(i-Bu)CO
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.