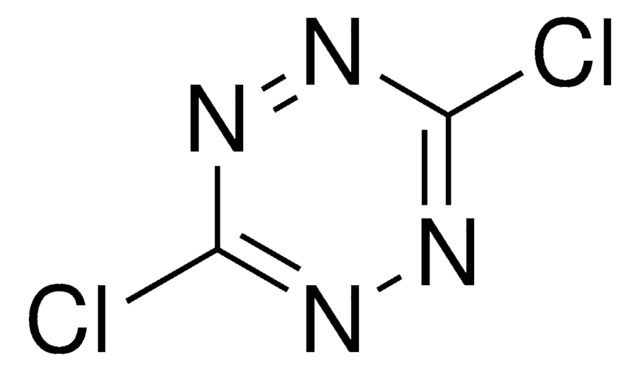
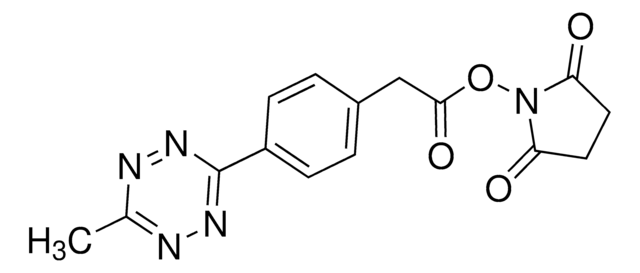
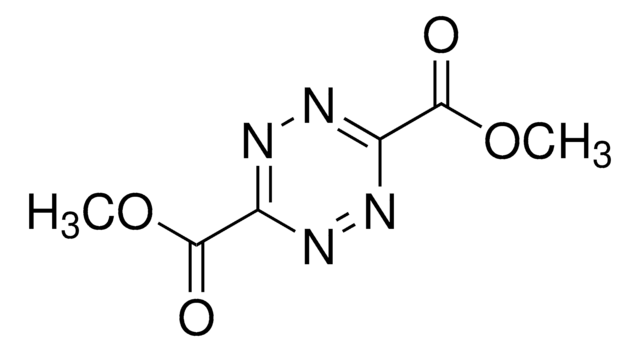
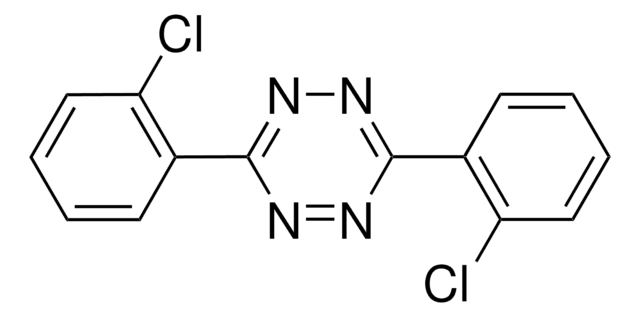
Wichtige Dokumente
ALD00108
3,6-Bis(methylthio)-1,2,4,5-tetrazine
≥95% (HPLC)
Synonym(e):
3,6-Bis(methylthio)-s-tetrazine, NSC 674352, s-Tetrazine
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥95% (HPLC)
Form
powder
mp (Schmelzpunkt)
82-87 °C
Lagertemp.
−20°C
SMILES String
CSC1=NN=C(SC)N=N1
InChI
1S/C4H6N4S2/c1-9-3-5-7-4(10-2)8-6-3/h1-2H3
InChIKey
ROUDTSAXQIBBFZ-UHFFFAOYSA-N
Anwendung
It can also used to synthesize:
- Substituted indoles and indolines derivatives by sequential [4+2] cycloaddition reactions.
- 3-(Methylthio)-6-(4-morpholinyl)-1,2,4,5-tetrazine by aromatic nucleophilic substitution reaction.
Ähnliches Produkt
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Die passende Version wird nicht angezeigt?
Wenn Sie eine bestimmte Version benötigen, können Sie anhand der Lot- oder Chargennummer nach einem spezifischen Zertifikat suchen.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Artikel
The inverse electron demand Diels-Alder reactions of electron-deficient heterocycles are significant cycloaddition reactions for the total synthesis of natural products containing highly substituted and functionalized heteroaromatic ring systems.
Verwandter Inhalt
As the exploration of the properties of complex natural products becomes increasingly more sophisticated with the technological advances being made in their screening and evaluation and as structural details of their interaction with biological targets becomes more accessible, the importance and opportunities for providing unique solutions to complex biological problems has grown. The Boger Lab addresses these challenging problems by understanding the complex solutions and subtle design elements that nature has provided in the form of a natural product and work to extend the solution through rational design elements to provide more selective, more efficacious, or more potent agents designed specifically for the problem or target under investigation. The resulting efforts have reduced many difficult or intractable synthetic challenges to manageable problems providing an approach not only to the natural product but one capable of simple extrapolation to a series of structural analogs with improved selectivity and efficacy.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.
![3-(([1,1′-Biphenyl]-4-ylmethyl)thio)-6-methyl-1,2,4,5-tetrazine >98%](/deepweb/assets/sigmaaldrich/product/structures/641/985/7d39c434-c9ea-490e-974f-7653ca3b9e8c/640/7d39c434-c9ea-490e-974f-7653ca3b9e8c.png)