754218
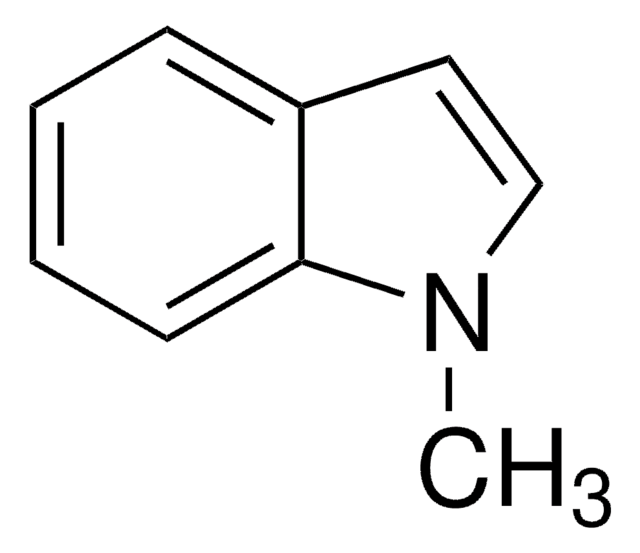
3,3-Dimethyl-1-(trifluormethyl)-1,2-Benziodoxol
98%
Synonym(e):
1,3-Dihydro-3,3-dimethyl-1-(trifluormethyl)-1,2-benziodoxol, Togni’s Reagenz
About This Item
Empfohlene Produkte
Assay
98%
Form
powder
mp (Schmelzpunkt)
75-79 °C
Lagertemp.
2-8°C
SMILES String
CC1(C)O[I](c2ccccc12)C(F)(F)F
InChI
1S/C10H10F3IO/c1-9(2)7-5-3-4-6-8(7)14(15-9)10(11,12)13/h3-6H,1-2H3
InChIKey
HVAPLSNCVYXFDQ-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Anwendung
- Selective trifluoromethylation of 1,3-disubstituted arenes through iridium-catalyzed arene borylation and copper-catalyzed trifluoromethylation
- Copper-catalyzed trifluoromethylation of aryl- and alkenylboronic acids with electrophilic trifluoromethylating reagent
- Pd-catalyzed electrophilic ortho-trifluoromethylation of arenes using trifluoroacetic acid as a promotor
Used in the Preparation of
- Trifluoromethylimidoylethyl substituted heterocycles via bis(trifluoromethylsulfonyl)amine-catalyzed Rotter type reaction of heterocycles with nitriles in presence of trifluoromethylbenziodoxole
- Stereoselective synthesis of α-trifluoromethyl aldehydes via trimethylbenzylimidazolidinone and copper-catalyzed enantioselective α-trifluoromethylation of aldehydes with iodonium salts
Ähnliches Produkt
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Flam. Sol. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
4.1B - Flammable solid hazardous materials
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Analysenzertifikate (COA)
Die passende Version wird nicht angezeigt?
Wenn Sie eine bestimmte Version benötigen, können Sie anhand der Lot- oder Chargennummer nach einem spezifischen Zertifikat suchen.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Verwandter Inhalt
Research in the Togni group focuses on the development of new ligands and reagents. These two general directions thus impact the ability to construct molecules in more efficient or unprecedented ways.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.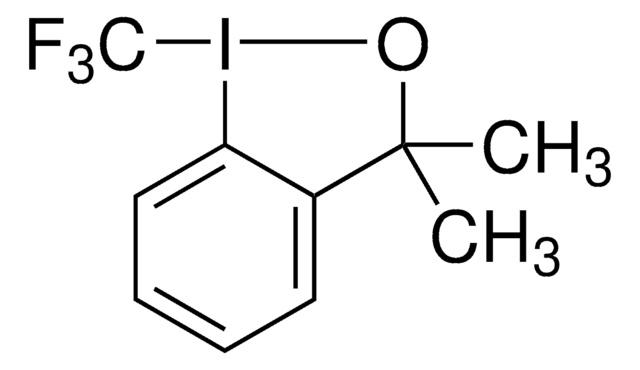



![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)