Alle Fotos(1)
Wichtige Dokumente
377198
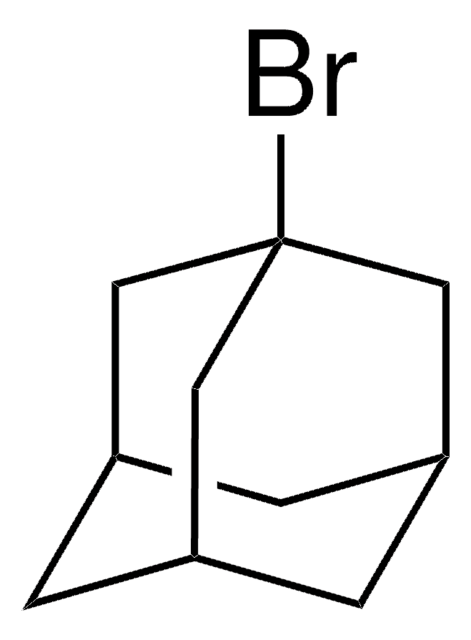
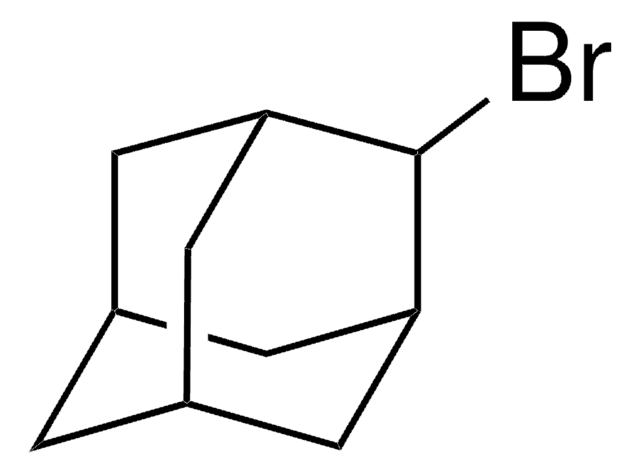
1-Iodadamantan
98%
Synonym(e):
1-Adamantyl iodide, 1-Iodotricyclo[3.3.1.13,7]decane, Adamantyl iodide
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C10H15I
CAS-Nummer:
Molekulargewicht:
262.13
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
98%
Form
solid
mp (Schmelzpunkt)
75-76 °C (lit.)
Funktionelle Gruppe
iodo
SMILES String
IC12C[C@H]3C[C@H](C[C@H](C3)C1)C2
InChI
1S/C10H15I/c11-10-4-7-1-8(5-10)3-9(2-7)6-10/h7-9H,1-6H2/t7-,8+,9-,10-
InChIKey
PXVOATXCSSPUEM-CHIWXEEVSA-N
Allgemeine Beschreibung
1-Iodoadamantane is a haloadamantane. Voltammetric reduction of 1-iodoadamantane at a silver cathode in tetrahydrofuran (THF) and acetonitrile (ACN) is reported to involve a single electron forming a mixture of monomeric and dimeric products. The photoinduced reaction of 1-iodoadamantane in DMSO is reported to afford substitution products on C3, C6, and C8, 1-adamantanol, 1-adamantyl 2-naphthyl ether, and adamantine. The photostimulated reaction of the phthalimide anion with 1-iodoadamantane is reported to yield 3-(1-adamantyl) phthalimide and 4-(1-adamantyl) phthalimide, along with the reduction product adamantane. 1-Iodoadamantane is reported to undergoe photostimulated reaction with the enolate anion of acetone, acetophenone and propiophenone to give admantane and the substitution products.
Anwendung
Employed in the free-radical carbonylation reactions with alkenes.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Photostimulated reaction of 1-iodoadamantane with carbanionic nucleophiles in DMSO by the SRN1 mechanism.
Borosky GL, et al.
The Journal of Organic Chemistry, 55(12), 3705-3707 (1990)
Juan E Argüello et al.
The Journal of organic chemistry, 68(6), 2362-2368 (2003-03-15)
The fluorescent excited state of the 2-naphthoxide ion (1) is quenched by aliphatic and aromatic halides according to an electron-transfer mechanism, with generation of the corresponding alkyl and aryl radicals by a concerted or consecutive C-X bond fragmentation reaction. Whereas
Dimerization of Cubene. 1-Iodoadamantane as a Probe for Radical Intermediates.
Lukin K and Eaton PE.
Journal of the American Chemical Society, 117(29), 7652-7656 (1995)
Rate study of haloadamantane reduction by samarium diiodide.
Lin T-Y, et al.
J. Chin. Chem. Soc., 49(6), 969-973 (2002)
Christopher A Paddon et al.
Ultrasonics sonochemistry, 14(5), 502-508 (2007-01-17)
The combination of ultrasound and electrochemistry -sonoelectrochemistry can produce a variety of effects within an electrochemical system including enhanced mass transport, in situ cleaning of an electrode surface, diminution of the diffusion layer, and possible induction of new reactions by
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.