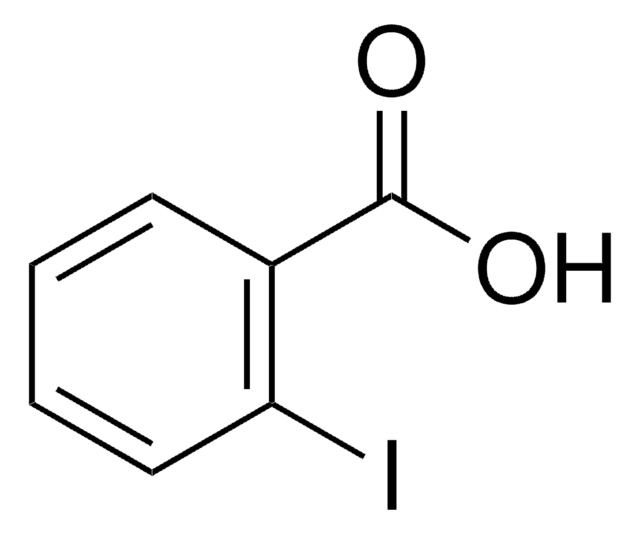
All Photos(1)
About This Item
Linear Formula:
C6H3Cl(I)COOH
CAS Number:
Molecular Weight:
282.46
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
163-167 °C (lit.)
SMILES string
OC(=O)c1ccc(Cl)cc1I
InChI
1S/C7H4ClIO2/c8-4-1-2-5(7(10)11)6(9)3-4/h1-3H,(H,10,11)
InChI key
LRRDANNSUCQNDU-UHFFFAOYSA-N
Related Categories
General description
4-Chloro-2-iodobenzoic acid is an electron-deficient substituted 2-iodobenzoic acid. It reacts with ynamide to provide the 3,4-disubstituted isocoumarin.
Application
4-Chloro-2-iodobenzoic acid may be used to synthesize:
- 6-chloro-3-(4-methoxyphenyl)-1H-isochromen-1-one
- (Z)-3-benzylidene-5-chloroisobenzofuran-1(3H)-one
- 6-chloro-3-pentyl-1H-isochromen-1-one
- 4-chloro-2-iodobenzophenone
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Regioselective synthesis of 3, 4-disubstituted isocoumarins through the Pd-catalyzed annulation of 2-iodoaromatic acids with ynamides.
Liu H, et al.
Chemical Communications (Cambridge, England), 52(41), 6801-6804 (2016)
Veronika Hampl et al.
Scientia pharmaceutica, 79(1), 21-30 (2011-05-28)
New isocoumarins were prepared in an efficient way from 2-iodobenzoic acid derivatives and hept-1-yne in a Sonogashira reaction, followed by spontaneous cyclization. Catalytic hydrogenation gave the corresponding dihydroisocoumarins. A 4-chloroisocoumarin was prepared on an alternative pathway. Some of the new
The synthesis and electronic absorption spectra of 3-phenyl-3 (4-pyrrolidino-2-substituted phenyl)-3H-naphtho [2, 1-b] pyrans: further exploration of the ortho substituent effect.
Gabbutt CD, et al.
Tetrahedron, 62(4), 737-745 (2006)
Regioselective One-Pot Synthesis of Isocoumarins and Phthalides from 2-Iodobenzoic Acids and Alkynes by Temperature Control.
Kumar MR, et al.
Advanced Synthesis & Catalysis, 355(16), 3221-3230 (2013)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service