All Photos(1)
About This Item
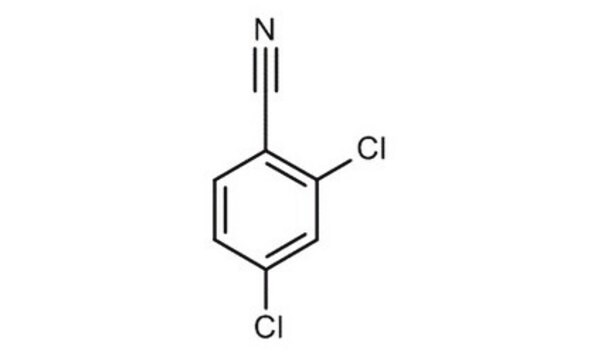
Linear Formula:
Cl2C6H3CN
CAS Number:
Molecular Weight:
172.01
Beilstein:
2326352
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
74-78 °C (lit.)
SMILES string
Clc1ccc(cc1Cl)C#N
InChI
1S/C7H3Cl2N/c8-6-2-1-5(4-10)3-7(6)9/h1-3H
InChI key
KUWBYWUSERRVQP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
3,4-Dichlorobenzonitrile (3,4-DCBN) is an aromatic nitrile that can be prepared from 3,4-dichlorobenzamide.
Application
3,4-Dichlorobenzonitrilemay be used in the preparation of:
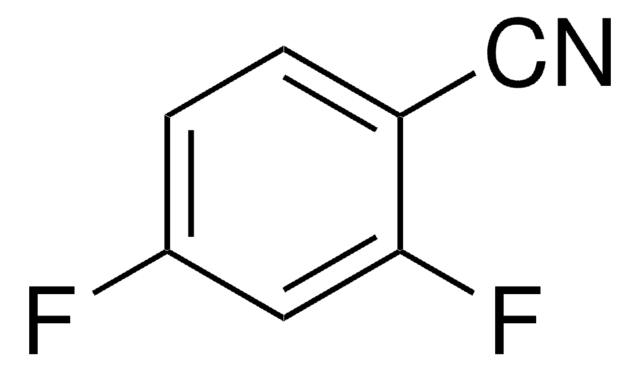
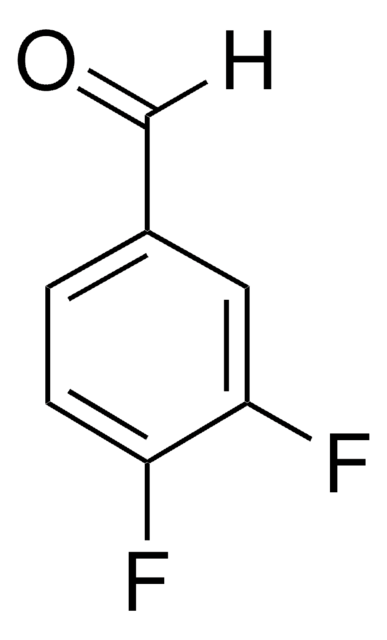
- 3,4-difluorobenzonitrile
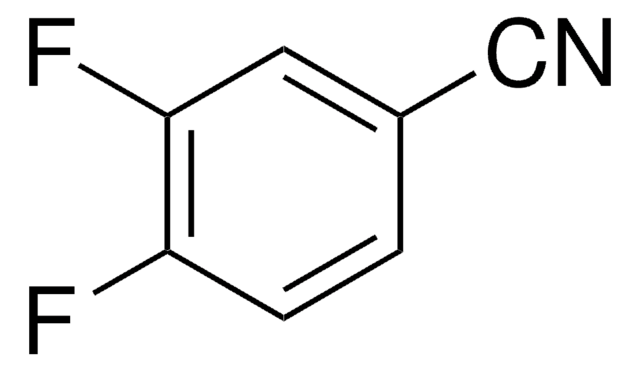
- 3-chloro-4-fluorobenzonitrile
- 5-chloromethyl-3-( 3,4-dichlorophenyl)isoxazole
- N-[3-(2-pyridyl)isoquinolin-1-yl]-3,4-dichlorobenzamidine
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis and copper-dependent antimycoplasmal activity of 1-amino-3-(2-pyridyl) isoquinoline derivatives. 2. Amidines.
De Zwart MAH, et al.
Journal of Medicinal Chemistry, 32(2), 487-493 (1989)
3-Aryl-5-halomethylisoxazoles. A new class of anthelmintics.
Sen HG, et al.
Journal of Medicinal Chemistry, 9(3), 431-433 (1966)
Synthesis of 3, 4-difluorobenzonitrile and monofluorobenzonitriles by means of halogen-exchange fluorination.
Suzuki H and Kimura Y.
Journal of Fluorine Chemistry, 52(3), 341-351 (1991)
Wouter Huiting et al.
eLife, 11 (2022-02-25)
A loss of the checkpoint kinase ataxia telangiectasia mutated (ATM) leads to impairments in the DNA damage response, and in humans causes cerebellar neurodegeneration, and an increased risk of cancer. A loss of ATM is also associated with increased protein
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service