515213
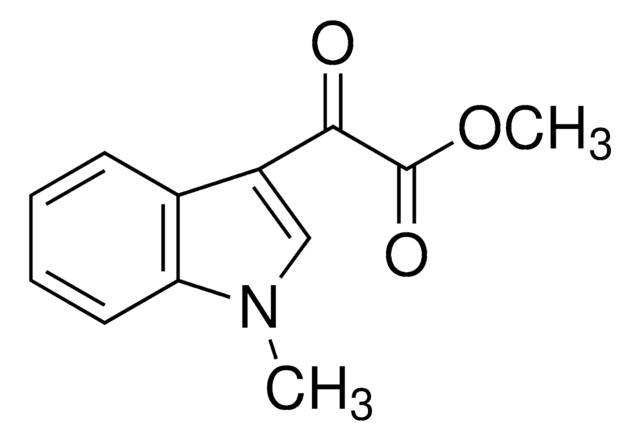
Methyl 3-indoleglyoxylate
98%
Synonym(s):
Methyl 2-(indol-3-yl)-2-oxoacetate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H9NO3
CAS Number:
Molecular Weight:
203.19
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
227-230 °C (lit.)
SMILES string
COC(=O)C(=O)c1c[nH]c2ccccc12
InChI
1S/C11H9NO3/c1-15-11(14)10(13)8-6-12-9-5-3-2-4-7(8)9/h2-6,12H,1H3
InChI key
VFIJGAWYVXDYLK-UHFFFAOYSA-N
Application
- Reactant for preparation of sotrastaurin analogs as protein kinase inhibitors
- Reactant for synthesis of GSK-3 inhibitors
- Reactant for Diels-Alder cycloaddition
- Reactant for preparation of a Janus kinase 3 inhibitor
- Reactant for synthesis of cephalandole alkaloids
- Reactant for stereoselective preparation of COX-2 inhibitor as anticancer agent
- Reactant for synthesis of cycloalkene indole carbazole alkaloids via ring-closing metathesis
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Manabu Nakazono et al.
Analytical sciences : the international journal of the Japan Society for Analytical Chemistry, 19(1), 123-127 (2003-02-01)
The chemiluminescence (CL) intensities of various indole derivatives substituted with a glyoxylyl group at the 3-position and a hydroxyl group at the 5-position of the indole ring were compared upon the addition of H2O2 in alkaline media. The CL intensities
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service