441627
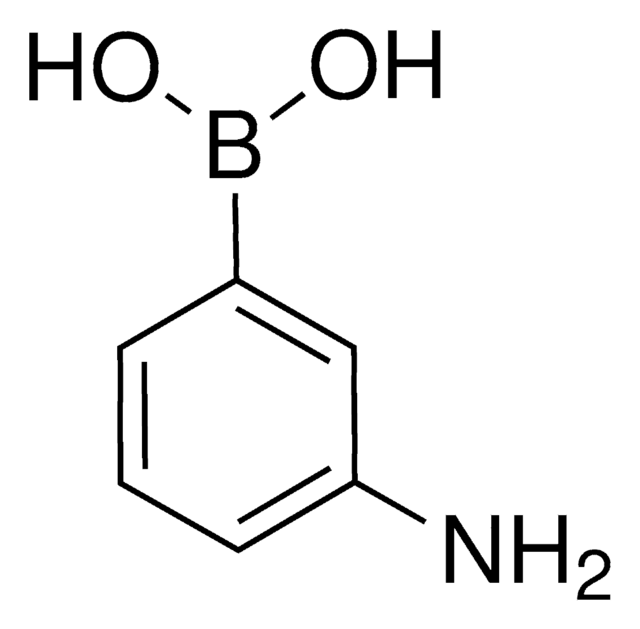
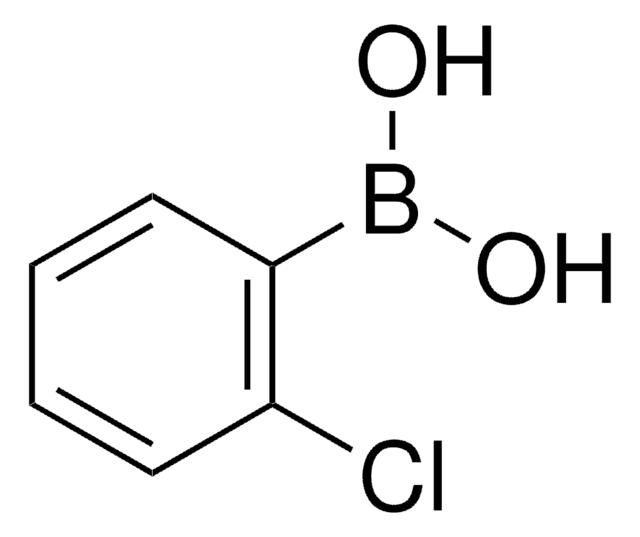
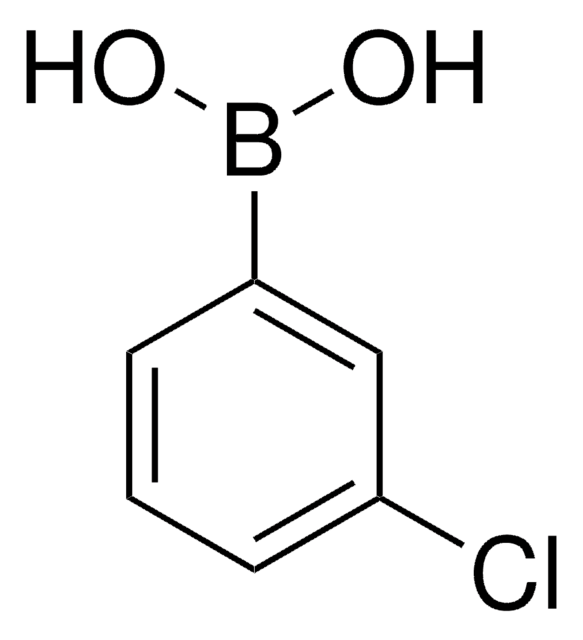
3-Bromophenylboronic acid
≥95%
Synonym(s):
3-Bromobenzeneboronic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
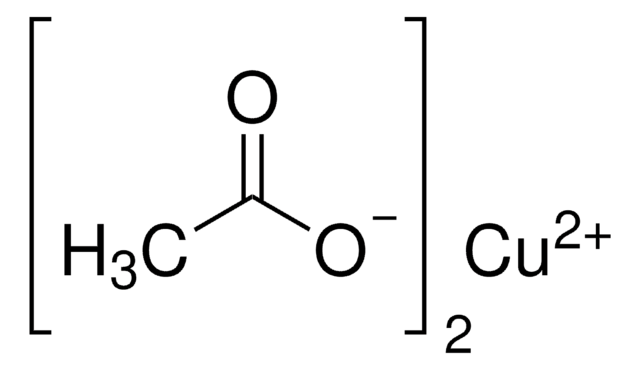
BrC6H4B(OH)2
CAS Number:
Molecular Weight:
200.83
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥95%
form
powder or crystals
mp
164-168 °C (lit.)
SMILES string
OB(O)c1cccc(Br)c1
InChI
1S/C6H6BBrO2/c8-6-3-1-2-5(4-6)7(9)10/h1-4,9-10H
InChI key
AFSSVCNPDKKSRR-UHFFFAOYSA-N
Application
Reactant involved in a variety of organic reactions including:
- Oxidative cross coupling
- Gold salt catalyzed homocoupling
- 1,4-Addition reactions with α,β-unsaturated ketones
- Enantioselective addition reactions
- Suzuki-Miyaura coupling for synthesis of anthranilamide-protected arylboronic acids
- C-H Functionalization of quinones
Other Notes
Contains varying amounts of anhydride
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G K Surya Prakash et al.
Organic letters, 6(13), 2205-2207 (2004-06-18)
[reaction: see text] A mixture of nitrate salt and chlorotrimethylsilane is found to be an efficient regioselective nitrating agent for the ipso-nitration of arylboronic acids to produce the corresponding nitroarenes in moderate to excellent yields. High selectivity, simplicity, and convenience
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)