All Photos(1)
About This Item
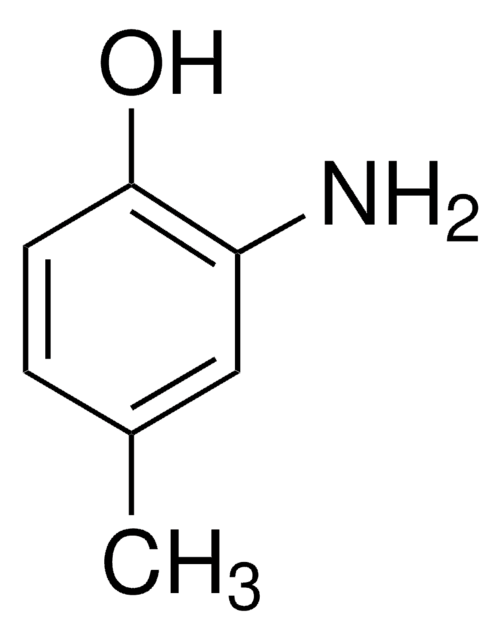
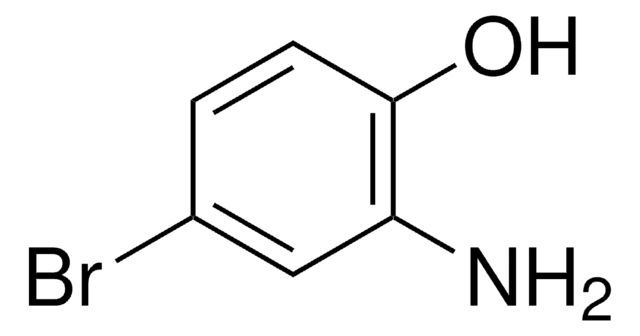
Linear Formula:
(CH3)3CC6H3(NH2)OH
CAS Number:
Molecular Weight:
165.23
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
160-163 °C (lit.)
SMILES string
CC(C)(C)c1ccc(O)c(N)c1
InChI
1S/C10H15NO/c1-10(2,3)7-4-5-9(12)8(11)6-7/h4-6,12H,11H2,1-3H3
InChI key
RPJUVNYXHUCRMG-UHFFFAOYSA-N
Related Categories
Application
2-Amino-4-tert-butylphenol can be used as a reactant to prepare:
- 4-tert-butyl-2-[(pyridylmethylene)amino]phenol intermediates, which are used to synthesize biologically important 2-(pyridyl)benzoxazole derivatives.
- Prolinamide phenols, as efficient hydrophobic organocatalysts for direct asymmetric aldol reaction aldehydes and ketones in water.
- N-(2-hydroxy-4-tert-butylphenyl)-acetamide, a key intermediate to prepare uranylsalophene derivatives which can be used as selective receptors in anion sensitive membrane sensors.
- Poly(2-amino-4-tert-butylphenol) [poly(2A-4TBP)] by electrochemical or chemical oxidative polymerization reaction.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
The chemical and electrochemical oxidative polymerization of 2-amino-4-tert-butylphenol
Abidi M, et al.
Electrochimica Acta, 212, 958-965 (2016)
Synthesis of 2-(2-, 3-, and 4-pyridyl) benzoxazoles by the reaction of phenolic Schiff bases with thianthrene cation radical.
Park MS, et al.
Journal of Heterocyclic Chemistry, 39(6), 1279-1282 (2002)
Rationally designed 4-phenoxy substituted prolinamide phenols organocatalyst for the direct aldol reaction in water.
Zhang S-P, et al.
Tetrahedron Letters, 50(11), 1173-1176 (2009)
Neutral anion receptors: synthesis and evaluation as sensing molecules in chemically modified field effect transistors.
Antonisse MMG, et al.
The Journal of Organic Chemistry, 62(26), 9034-9038 (1997)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![4-(DIMETHYLAMINO)BENZALDEHYDE [4-(DIMETHYLAMINO)BENZYLIDENE]HYDRAZONE AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/268/291/5232c253-7dd7-435c-b094-6d334239d9fb/640/5232c253-7dd7-435c-b094-6d334239d9fb.png)