900402
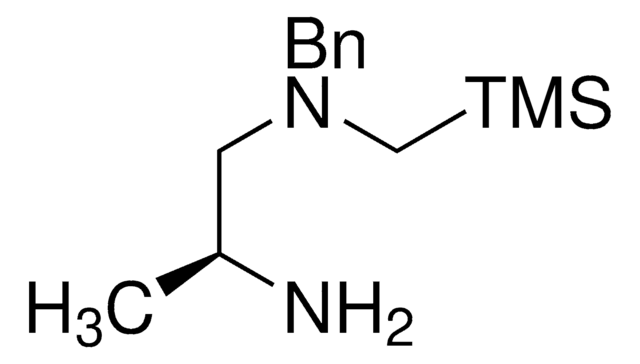
SLAP HydroQuinoxaline
95%
Synonym(s):
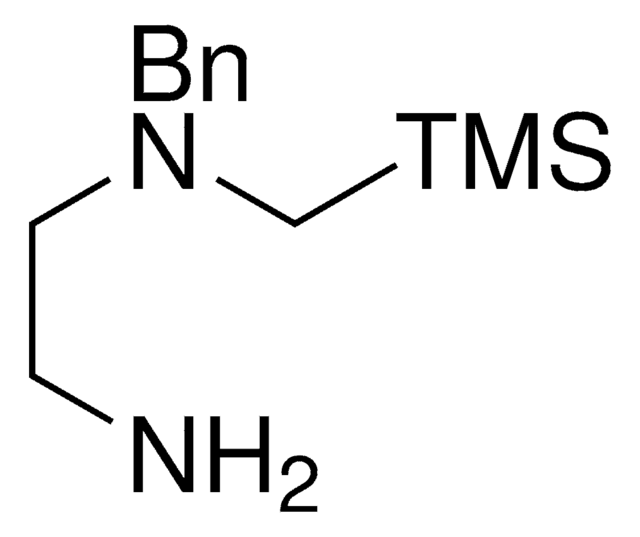
trans-N1-Benzyl-N1-((trimethylsilyl)methyl)cyclohexane-1,2-diamine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C17H30N2Si
Molecular Weight:
290.52
UNSPSC Code:
12352200
NACRES:
NA.22
Recommended Products
Assay
95%
form
liquid
refractive index
n/D 1.5222
density
0.961 g/mL
storage temp.
−20°C
Application
Silicon amine protocol (SLAP) reagents, in conjunction with aldehydes and ketones, form N-unprotected piperazines via photocatalytic cross-coupling with Ir[(ppy)2dtbbpy]PF6 (747769). Heteroaromatic, aromatic, and aliphatic aldehydes are well tolerated, as are structurally and stereochemically complex SLAP reagents. This product was introduced in collaboration with the Bode Research Group and provides a tin-free alternative to SnAP (tin amine protocol) reagents for piperazine synthesis.
Other Notes
- Technology Spotlight: SLAP Reagents for Piperazine Synthesis
- Silicon Amine Reagents for the Photocatalytic Synthesis of Piperazines from Aldehydes and Ketones
- Lewis Acid Induced Toggle from Ir(II) to Ir(IV) Pathways in Photocatalytic Reactions: Synthesis of Thiomorpholines and Thiazepanes from Aldehydes and SLAP Reagents.
- Continuous Flow Synthesis of Morpholines and Oxazepanes with Silicon Amine Protocol (SLAP) Reagents and Lewis Acid Facilitated Photoredox Catalysis
related product
Product No.
Description
Pricing
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
230.0 °F
Flash Point(C)
> 110 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Moritz K Jackl et al.
Organic letters, 19(17), 4696-4699 (2017-08-17)
Photocatalytic coupling of aldehydes and silicon amine protocol (SLAP) reagents enables the simple, scalable synthesis of substituted morpholines, oxazepanes, thiomorpholines, and thiazepanes under continuous flow conditions. Key to the success of this process is the combination of an inexpensive organic
Sheng-Ying Hsieh et al.
Organic letters, 18(9), 2098-2101 (2016-04-22)
Silicon amine protocol (SLAP) reagents for photocatalytic cross-coupling with aldehydes and ketones to form N-unprotected piperazines have been developed. This blue light promoted process tolerates a wide range of heteroaromatic, aromatic, and aliphatic aldehydes and structurally and stereochemically complex SLAP
Sheng-Ying Hsieh et al.
ACS central science, 3(1), 66-72 (2017-02-06)
Redox neutral photocatalytic transformations often require careful pairing of the substrates and photoredox catalysts in order to achieve a catalytic cycle. This can limit the range of viable transformations, as we recently observed in attempting to extend the scope of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
iridium(III) 97%](/deepweb/assets/sigmaaldrich/product/structures/309/053/0823f035-245c-433d-b033-2eca2d931c67/640/0823f035-245c-433d-b033-2eca2d931c67.png)