290734
Dabco® 33-LV
Synonym(s):
1,4-Diazabicyclo[2.2.2]octane solution
About This Item
Recommended Products
description
33 wt. % solution of triethylenediamine in dipropylene glycol
density
1.02 g/mL
SMILES string
C1CN2CCN1CC2
InChI
1S/C6H12N2/c1-2-8-5-3-7(1)4-6-8/h1-6H2
InChI key
IMNIMPAHZVJRPE-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Dabco®33-LV (Db) is a gelling catalyst and a bidentate ligand that forms a self-assembled monolayer (SAM) on a variety of substrates. It functionalizes the surface and immobilizes the surface atoms.
Application
- As a gel catalyst to promote the gel reaction in the production of flexible and rigid polyurethane foams, microcellular foams, coatings, and elastomers. It is used to accelerate the isocyanate-polyol reaction.
- In the synthesis of anion exchange membranes (AEMs) via electrospinning, contributing to the crosslinking between the polymers and the mechanical stability of the membrane. AEMs are further used in fuel cells, water electrolysis, redox flow batteries, and electrolyzers.
- Db can be used in the preparation of epoxy based soybean oil for the formation of polyurethane foams.
Legal Information
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Based on the same working principle as the nontraceless Staudinger Ligation the auxiliary phosphine reagent can be cleaved from the product after the ligation is completed leaving a native amide bond. Thus, the total chemical synthesis of proteins and glycopeptides is enabled overcoming the limitations of native chemical ligation (NCL) of a Cys residue at the ligation juncture.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)
![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)
![1,4-Diazabicyclo[2.2.2]octane for synthesis](/deepweb/assets/sigmaaldrich/product/images/488/587/f5a877b3-e573-4686-931f-648015f4d284/640/f5a877b3-e573-4686-931f-648015f4d284.jpg)

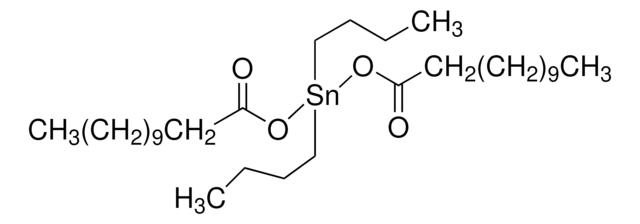
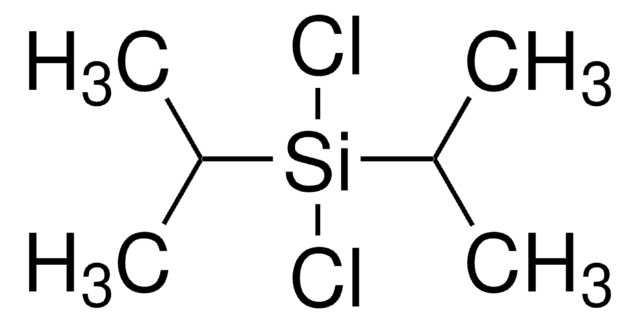
![1,5,7-Triazabicyclo[4.4.0]dec-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/171/446/333d560c-cff6-4958-b489-5acfb3057cce/640/333d560c-cff6-4958-b489-5acfb3057cce.png)


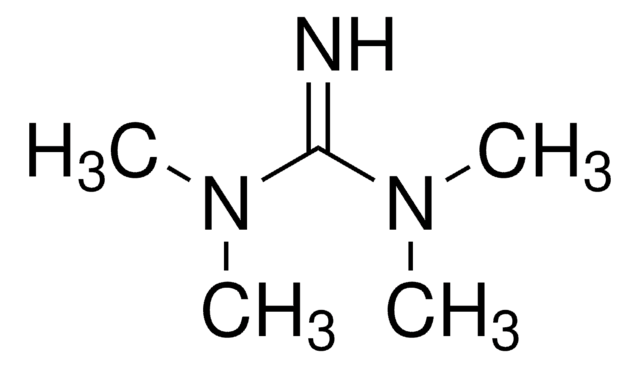

![1,5-Diazabicyclo[4.3.0]non-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/400/401/859b2474-712b-4448-b231-74d0bc3203f1/640/859b2474-712b-4448-b231-74d0bc3203f1.png)
![Bis(trimethylaluminum)-1,4-diazabicyclo[2.2.2]octane adduct](/deepweb/assets/sigmaaldrich/product/structures/978/293/6c8c7fbe-4b40-4576-bd40-94ac58cbe057/640/6c8c7fbe-4b40-4576-bd40-94ac58cbe057.png)