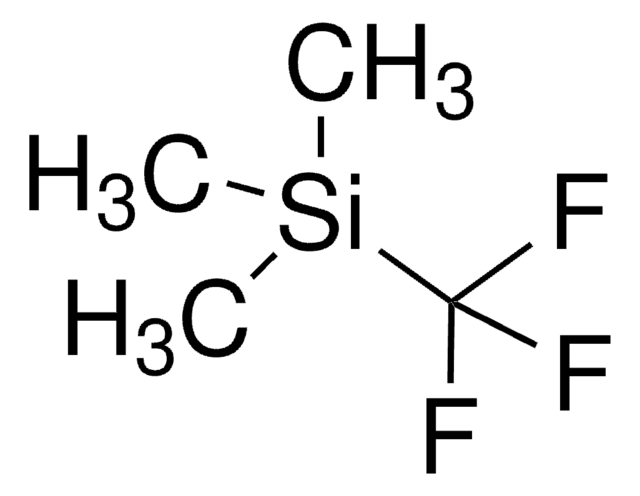
248061
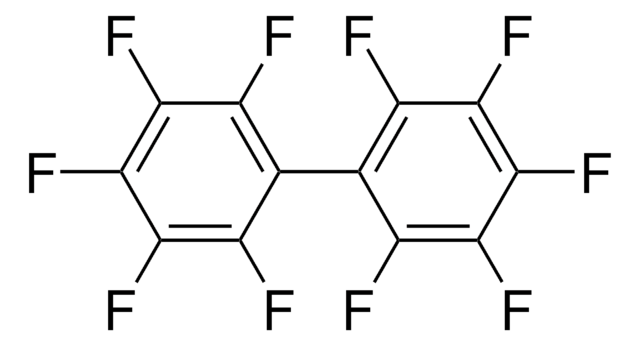
Octafluoronaphthalene
96%
Synonym(s):
1,2,3,4,5,6,7,8-Octafluoronaphthalene, Perfluoronaphthalene
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10F8
CAS Number:
Molecular Weight:
272.09
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
96%
form
solid
mp
87-88 °C (lit.)
SMILES string
Fc1c(F)c(F)c2c(F)c(F)c(F)c(F)c2c1F
InChI
1S/C10F8/c11-3-1-2(5(13)9(17)7(3)15)6(14)10(18)8(16)4(1)12
InChI key
JDCMOHAFGDQQJX-UHFFFAOYSA-N
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Amination of octafluoronaphthalene in liquid ammonia: 2, 6-and 2, 7-Diaminohexafluoronaphthalenes selective preparation.
Journal of Fluorine Chemistry, 129(4), 253-260 (2008)
Qimeng Jiang et al.
Polymers, 12(7) (2020-07-28)
Solid acid catalysts generally show the disadvantage of low acid amount and low recycling rate. To solve these problems, corn stalk-based solid acid catalysts were synthesized through carbonization and sulfonation processes in this work. The results showed that besides the
J C Collings et al.
Acta crystallographica. Section C, Crystal structure communications, 57(Pt 7), 870-872 (2001-07-10)
The structure of the title complex, C10F8*C14H10, comprises mixed stacks of alternating diphenylacetylene and octafluoronaphthalene molecules, both lying at inversion centres and parallel to within 8.6 (1) degrees, in contrast with the herring-bone packing observed in crystals of either pure
A phase transformation with no change in space group symmetry; octafluoronaphthalene.
Pawley GS and Dietrich O-W.
Journal of Physics C: Solid State Physics, 8(16), 2549-2549 (1975)
Polycyclic fluoroaromatic compounds. Part XII.[1] The extent of 1-substitution in octafluoronaphthalene.
Burdon J and Rimmington TW.
Journal of Fluorine Chemistry, 27(3), 257-261 (1985)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service




![Sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate](/deepweb/assets/sigmaaldrich/product/structures/251/439/7a621e74-bfd1-4a43-833c-09adfcc1e0b3/640/7a621e74-bfd1-4a43-833c-09adfcc1e0b3.png)