SMB00609
6,6′-Dihydroxythiobinupharidine
≥95% (HPLC)
Sinónimos:
6,6′-Dihydroxythionuphlutine A, Nuphleine
About This Item
Productos recomendados
Quality Level
assay
≥95% (HPLC)
form
powder
antibiotic activity spectrum
Gram-positive bacteria
mode of action
enzyme | inhibits
storage temp.
−20°C
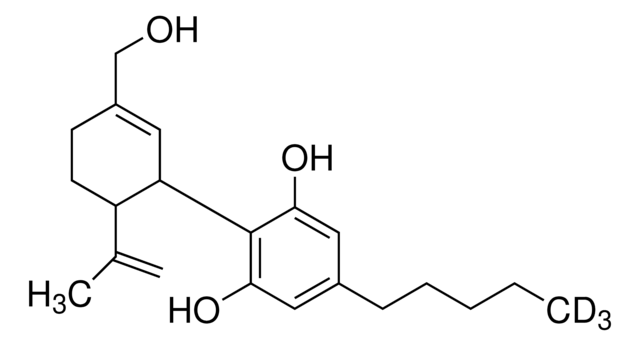
SMILES string
O[C@H]1N2[C@@]([C@H](C)CC[C@H]2C3=COC=C3)([H])CC[C@@]14SC[C@]5(C4)CC[C@]([C@H](C)CC[C@H]6C7=COC=C7)([H])N6[C@@H]5O
InChI
1S/C30H42N2O4S/c1-19-3-5-25(21-9-13-35-15-21)31-23(19)7-11-29(27(31)33)17-30(37-18-29)12-8-24-20(2)4-6-26(32(24)28(30)34)22-10-14-36-16-22/h9-10,13-16,19-20,23-28,33-34H,3-8,11-12,17-18H2,1-2H3/t19-,20-,23+,24+,25+,26+,27-,28-,29-,30+/m1/s1
InChI key
DYEOLAMWQVWASS-XKCSGWQSSA-N
General description
Biochem/physiol Actions
It was also found to act synergistically with cytotoxic drugs such as cisplatin and etoposide, enabling their cytotoxic effect at lower concentrations.
6,6′-Dihydroxythiobinupharidine was found to have cytotoxic activity at a concentration of ~10 μM on human leukemia cells (U937), mouse melanoma cells (B16F10), and human fibroblasts (HT1080).
In addition, Nuphar lutea extract was effective against both Leishmania promastigote and amastigote forms (IC50 = 2 ± 0.12 μg/mL; ID50 = 0.65 ± 0.023 μg/mL; LD50 = 2.1 ± 0.096 μg/mL, STI = 3.23). A synergistic antileishmanial activity was demonstrated with the antileishmanial drug, paromomycin.
Recently 6,6′-dihydroxythiobinupharidine was found to be active against MRSA and VRE strains with an MIC of 1-4 μg/mL. Inhibition of DNA topoisomerase IV but not DNA gyrase in S. aureus was suggested as the mechanism of action. 6,6′-Dihydroxythiobinupharidine was also shown to promote neutrophil effector bactericidal functions.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico