SMB00089
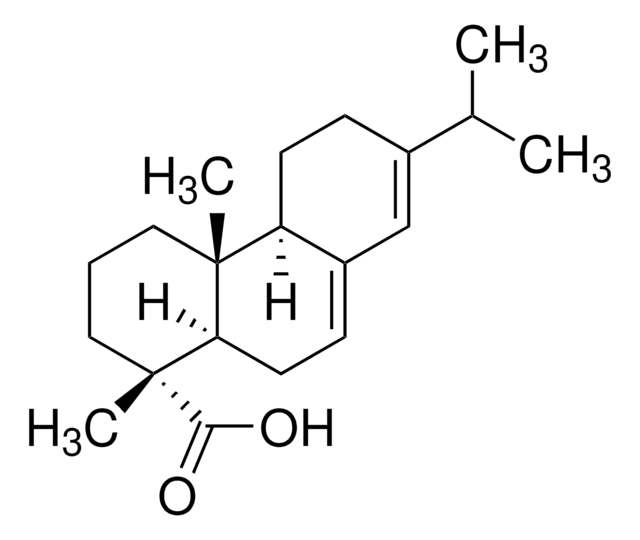
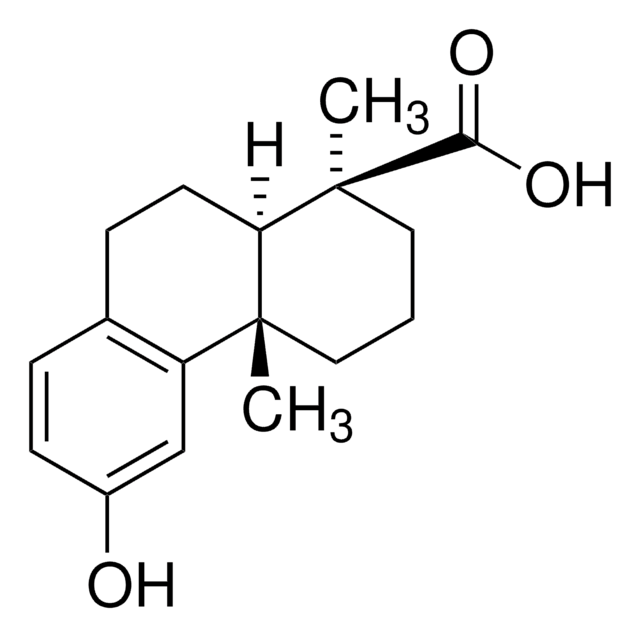
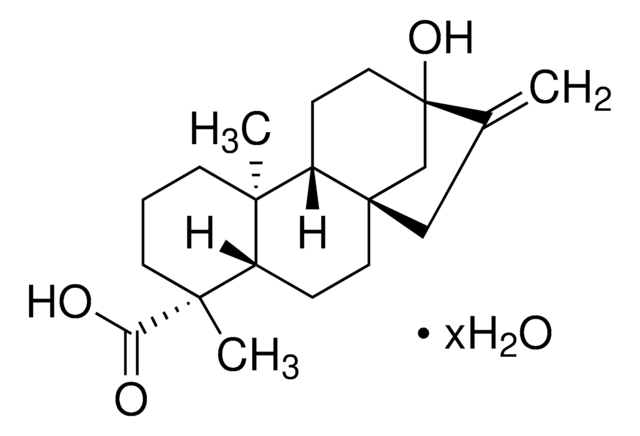
Dehydroabietic acid
≥90% (LC/MS-ELSD)
Sinónimos:
Abieta-8,11,13-trien-18-oic acid, Dehydroabietate
About This Item
Productos recomendados
assay
≥90% (LC/MS-ELSD)
form
solid
application(s)
metabolomics
vitamins, nutraceuticals, and natural products
storage temp.
−20°C
SMILES string
CC(C)c1ccc2c(CC[C@H]3[C@@](C)(CCC[C@]23C)C(O)=O)c1
InChI
1S/C20H28O2/c1-13(2)14-6-8-16-15(12-14)7-9-17-19(16,3)10-5-11-20(17,4)18(21)22/h6,8,12-13,17H,5,7,9-11H2,1-4H3,(H,21,22)/t17-,19-,20-/m1/s1
InChI key
NFWKVWVWBFBAOV-MISYRCLQSA-N
Categorías relacionadas
General description
Application
- to study the aging process of Pinus resins using Fourier-transform infrared spectroscopy (FTIR)
- to study the composition changes in Pinus genus with aging using Raman spectroscopy complemented with infrared spectroscopy
- to estimate resistance against biotic stress as proxy in chemical defenses in Pinus halepensis
Biochem/physiol Actions
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Global Trade Item Number
| Número de referencia del producto (SKU) | GTIN |
|---|---|
| SMB00089-1MG | 4061837076176 |
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico