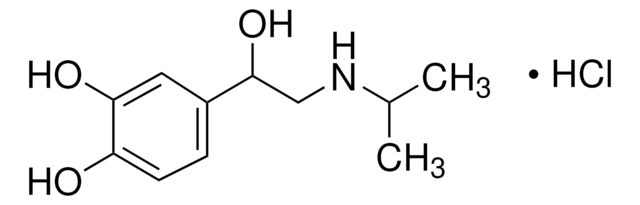
M3668
Metergoline
Sinónimos:
N-CBZ-[(8β)-1,6-Dimethylergolin-8-yl]methylamine, [(8β)-1,6-Dimethylergolin-8-yl)methyl]carbamic acid phenylmethyl ester
About This Item
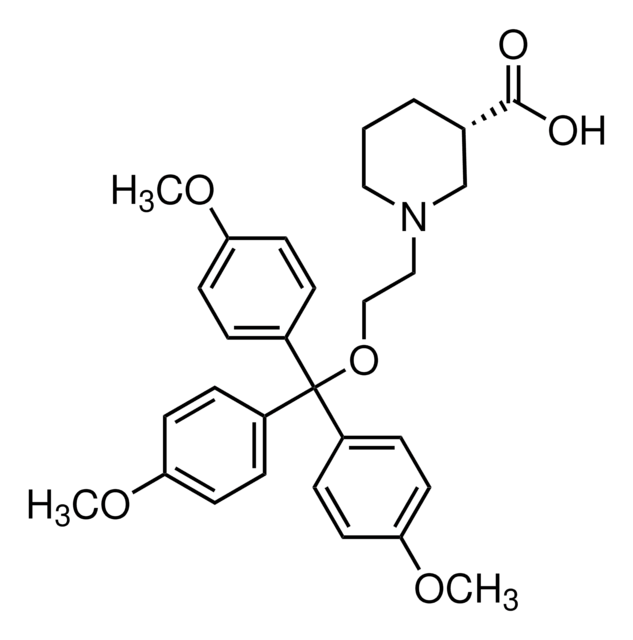
Productos recomendados
mp
148-150 °C (lit.)
solubility
0.1 M HCl: 1.4 mg/mL
ethanol: 4 mg/mL
H2O: insoluble
storage temp.
−20°C
SMILES string
[H][C@]2(CNC(=O)OCc1ccccc1)CN(C)[C@]3([H])Cc4cn(C)c5cccc(c45)[C@@]3([H])C2
InChI
1S/C25H29N3O2/c1-27-14-18(13-26-25(29)30-16-17-7-4-3-5-8-17)11-21-20-9-6-10-22-24(20)19(12-23(21)27)15-28(22)2/h3-10,15,18,21,23H,11-14,16H2,1-2H3,(H,26,29)/t18-,21+,23+/m0/s1
InChI key
WZHJKEUHNJHDLS-QTGUNEKASA-N
Gene Information
human ... HTR1A(3350) , HTR1B(3351) , HTR1D(3352) , HTR1E(3354) , HTR1F(3355) , HTR2A(3356) , HTR2B(3357) , HTR2C(3358) , HTR7(3363)
rat ... Htr7(65032)
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Application
- to study its effects on astrocyte calcium signals evoked by whisker stimulation
- to study its effects on lipopolysaccharide-induced anorexia in rats
- to evaluate the non-specific binding by 5-HT1A receptor binding assays
Biochem/physiol Actions
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico