CMP472TS1
Milligard® PES 1,2/0,45 µm, Cartucho
pore size 1.2/0.45 μm, cartridge nominal length 20 in. (50 cm)
Sinónimos:
Milligard PES 20" 1.2/0.45 μm Cartridge
About This Item
Productos recomendados
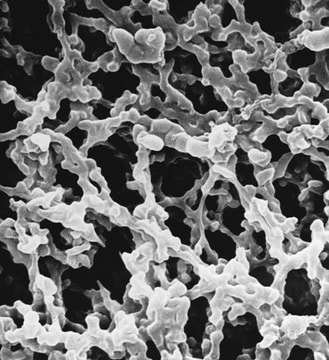
material
polyethersulfone membrane
polypropylene housing
polypropylene support
polypropylene vent cap
Quality Level
reg. compliance
meets FDA Indirect Food Additive requirements cited in 21 CFR 177-182 (all component materials)
sterility
non-sterile
sterilization compatibility
autoclavable compatible
steam-in-place compatible
product line
EMPROVE® Filter
Milligard®
feature
hydrophilic
manufacturer/tradename
Milligard®
parameter
1.38 bar max. differential pressure (20 psid) at 25 °C (Reverse)
5.52 bar max. differential pressure (80 psid) at 4-25 °C (Forward)
technique(s)
prefiltration: suitable
L
22.8 in.
W
2.7 in.
cartridge nominal length
20 in. (50 cm)
diam.
69 mm (2.71 in.)
filtration area
1.20 m2
impurities
<0.25 EU/mL bacterial endotoxins (LAL test, per device; aqueous extraction; meeting requirements of USP <85>, EP 2.6.14 and JP 4.01)
matrix
Milligard® PES
pore size
1.2/0.45 μm pore size
input
sample type liquid
cartridge code
Code 7
General description
Other Notes
- Organism Retention: Microorganism
- Mode of Action: Filtration (size exclusion)
- Application: BioProcessing
- Intended Use: Reduction or removal of microorganism/bioburden
- Instructions for Use: Please refer wetting instructions of Filters with MILLIGARD® PES Membrane user guide
- Storage Statement: Please refer Filters with MILLIGARD® PES Membrane user guide
- Disposal Statement: Dispose of in accordance with applicable federal, state and local regulations.
Preparation Note
Autoclave wet 25x, 60 minute cycles at 128°C. In-line steam (wet): 10 forward cycles, 30 min.,
135 °C at ≤0.34 bar (5 psid) and 5 reverse cycles,
30 min., 135 °C at ≤0.07 bar (<1 psid)*.
*Devices were validated using steam at full differential pressure starting at 31 psid to a final differential pressure of 1 psid, resulting in a calculated average of 4 psid in forward and reverse directions.
Legal Information
¿No encuentra el producto adecuado?
Pruebe nuestro Herramienta de selección de productos.
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico