M5852
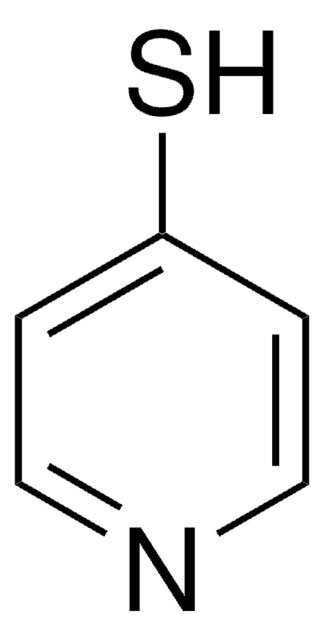
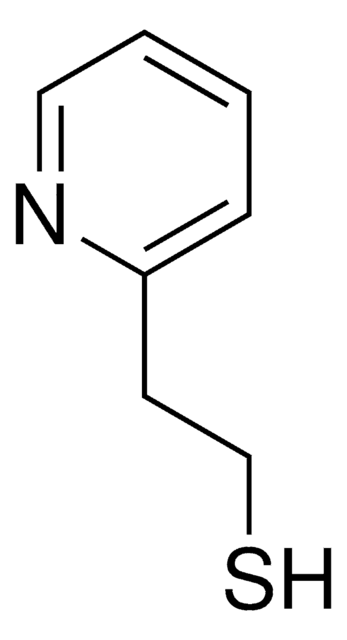
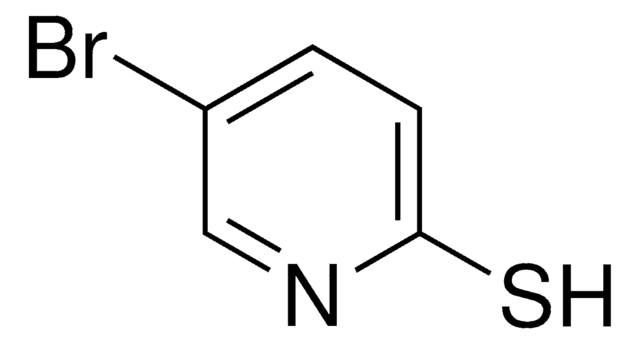
2-Mercaptopyridine
ReagentPlus®, 99%
Sinónimos:
2-Pyridinethiol, 2-Pyridyl mercaptan
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C5H5NS
Número de CAS:
Peso molecular:
111.16
Beilstein/REAXYS Number:
105787
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
product line
ReagentPlus®
assay
99%
mp
127-130 °C (lit.)
storage temp.
2-8°C
SMILES string
Sc1ccccn1
InChI
1S/C5H5NS/c7-5-3-1-2-4-6-5/h1-4H,(H,6,7)
InChI key
WHMDPDGBKYUEMW-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
2-Mercaptopyridine is an organosulfur compound that contains more than one hetero atom. It is commonly used as a nucleophile in various organic synthesis reactions and plays important role in coordination chemistry as a versatile ligand due to its π-acidic nature.
Application
Employed as a ligand in metal complexes.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Étienne Rochette et al.
Journal of the American Chemical Society, 141(31), 12305-12311 (2019-07-10)
The potential advantages of using arylboronic esters as boron sources in C-H borylation are discussed. The concept is showcased using commercially available 2-mercaptopyridine as a metal-free catalyst for the transfer borylation of heteroarenes using arylboronates as borylation agents. The catalysis
Chinese Chemical Letters = Zhongguo Hua Xue Kuai Bao, 3, 741-741 (1992)
Partha Halder et al.
Inorganic chemistry, 50(3), 708-710 (2010-12-25)
Iron(II) tris(2-pyridylthio)methanido (1) containing an Fe-C bond, obtained from the reaction of tris(2-pyridylthio)methane (HL(1)) and iron(II) triflate, reacts with protic acid to generate iron(II) bis(2-pyridylthio)carbene (1a). The carbene complex is converted to an iron(II) complex (2) of the 1-[bis(2-pyridylthio)methyl]pyridine-2-thione ligand
Alexey V Chuchuryukin et al.
Inorganic chemistry, 45(5), 2045-2054 (2006-02-28)
A silver-free route has been employed for the synthesis of a number of Pd and Pt complexes supported by an NCN "pincer" ligand (NCN = [2,6-(Me2NCH2)2C6H3]-) via halide abstraction. This was achieved by the use of o-, m-, and p-hydroxypyridines
H I Abdulla et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 57(13), 2659-2671 (2002-01-05)
The infrared spectra of 2-hydroxypyridine (2-OHP), 2-thiopyridine (2-SHP), and 2-aminopyridine (2-NH2P) have been recorded in the solid, liquid and vapor phases in the region 4000-200 cm(-1). To support the work, deuterated forms of these compounds in the functional groups XH
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico