857459
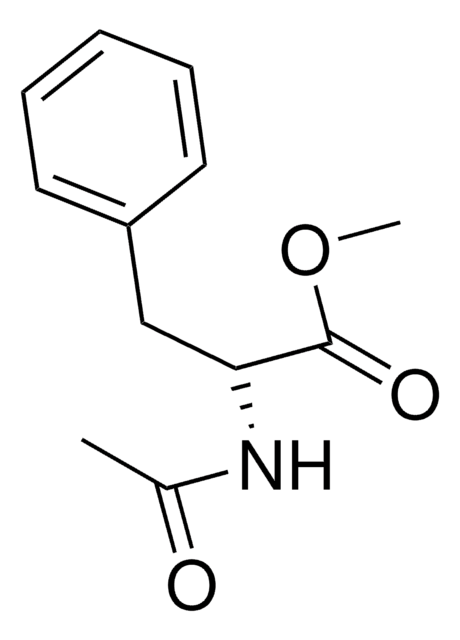
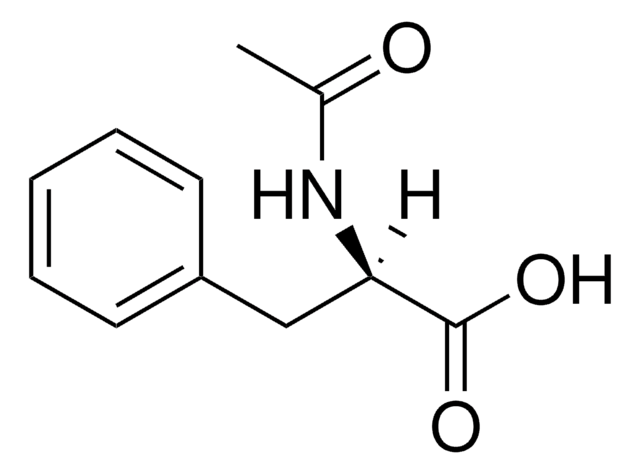
N-Acetyl-L-phenylalanine
ReagentPlus®, 99%
Sinónimos:
(+)-N-Acetylphenylalanine, (S)-2-Acetamido-3-phenylpropanoic acid
About This Item
Productos recomendados
Quality Level
product line
ReagentPlus®
assay
99%
form
powder
optical activity
[α]22/D +40.0°, c = 1 in methanol
reaction suitability
reaction type: C-H Activation
reaction type: solution phase peptide synthesis
reagent type: ligand
reaction type: Peptide Synthesis
mp
171-173 °C (lit.)
application(s)
peptide synthesis
functional group
amine
carboxylic acid
SMILES string
CC(=O)N[C@@H](Cc1ccccc1)C(O)=O
InChI
1S/C11H13NO3/c1-8(13)12-10(11(14)15)7-9-5-3-2-4-6-9/h2-6,10H,7H2,1H3,(H,12,13)(H,14,15)/t10-/m0/s1
InChI key
CBQJSKKFNMDLON-JTQLQIEISA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
- N-acetyl phenylalanine methyl ester by esterification reaction with methanol using Mukaiyama′s reagent.
- Acetylaminocyclohexane propanoic acid by rhodium-catalyzed hydrogenation reaction.
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Chromatograms
application for HPLCNuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico