557749
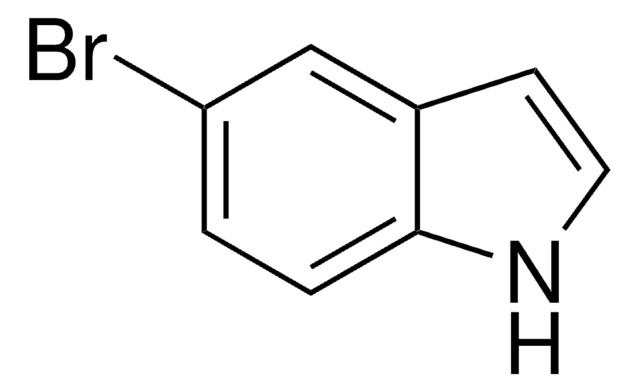
N-Boc-5-bromoindole
97%
Sinónimos:
tert-Butyl 5-bromoindole-1-carboxylate
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C13H14BrNO2
Número de CAS:
Peso molecular:
296.16
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
97%
form
solid
mp
56-57 °C (lit.)
functional group
bromo
SMILES string
CC(C)(C)OC(=O)n1ccc2cc(Br)ccc12
InChI
1S/C13H14BrNO2/c1-13(2,3)17-12(16)15-7-6-9-8-10(14)4-5-11(9)15/h4-8H,1-3H3
InChI key
PBWDRTGTQIXVBR-UHFFFAOYSA-N
Categorías relacionadas
General description
N-Boc-5-bromoindole can undergo Sonogashira coupling reaction with N,N-diisopropylprop-2-ynylamine to afford the corresponding propargylic diisopropylamine. N-Boc-5-bromoindole is formed as an intermediate during the synthesis of 2-(5-substituted-1H-indol-3-yl)-N-hydroxyacetamide derivatives.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Hiroyuki Nakamura et al.
The Journal of organic chemistry, 70(6), 2357-2360 (2005-03-12)
[reaction: see text] Propargylic diisopropylamines containing heterocycles, which were prepared readily from heterocyclic bromides and propargyldiisopropylamine by the Sonogashira coupling reaction, underwent the allene transformation reaction in the presence of Pd(2)(dba)(3).CHCl(3) catalyst (2.5 mol %) and 1,2-bis[bis(pentafluorophenyl)phosphino]ethane (10 mol %)
Sylvain Petit et al.
ChemMedChem, 4(2), 261-275 (2008-12-05)
The lead compound 5-bromoindolyl-3-acetohydroxamic acid (10) was recently identified as a potent inhibitor of bacterial peptide deformylases (PDFs). The synthesis and associated activities of new variants were investigated at position 5 to optimize the fit at the S1' subsite and
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico