425664
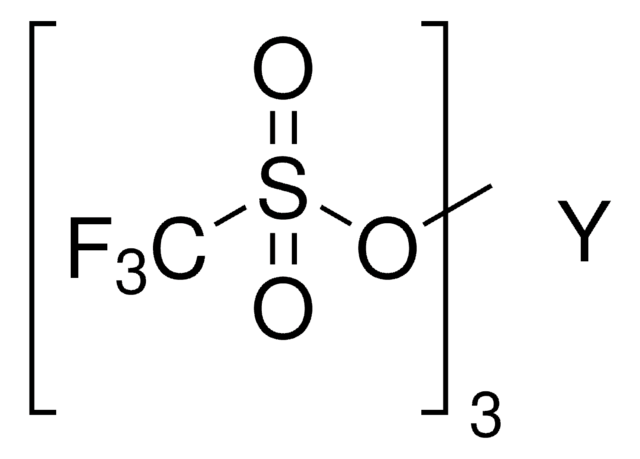
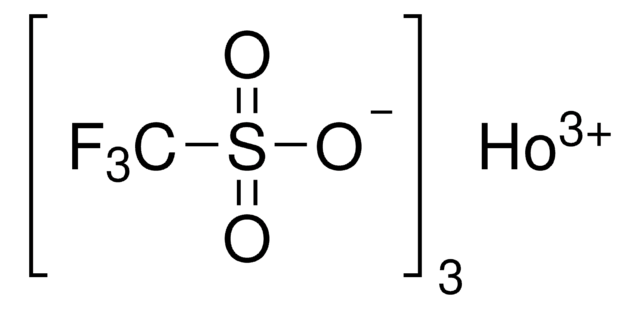
Dysprosium(III) trifluoromethanesulfonate
98%
Sinónimos:
Dysprosium(III) triflate, Tris(triflato)dysprosium
About This Item
Productos recomendados
assay
98%
reaction suitability
core: dysprosium
reagent type: catalyst
reaction type: Ring-Opening Polymerization
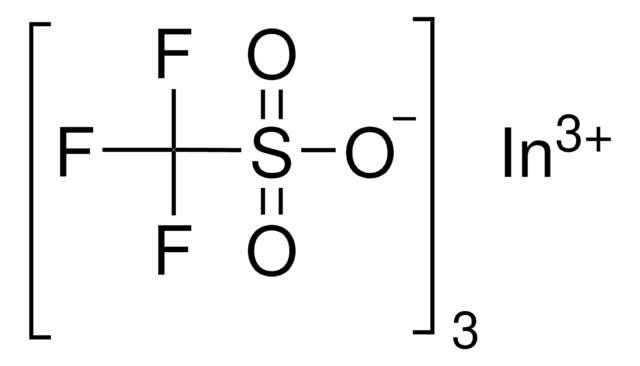
SMILES string
[Dy+3].[O-]S(=O)(=O)C(F)(F)F.[O-]S(=O)(=O)C(F)(F)F.[O-]S(=O)(=O)C(F)(F)F
InChI
1S/3CHF3O3S.Dy/c3*2-1(3,4)8(5,6)7;/h3*(H,5,6,7);/q;;;+3/p-3
InChI key
XSVCYDUEICANRJ-UHFFFAOYSA-K
Categorías relacionadas
General description
Application
- Aza-Piancatelli rearrangement
- Friedel-Crafts alkylation
- Ring-opening polymerization reactions
- Microwave-assisted Kabachnik-Fields condensation
- Cycloaddition reactions (Lewis-acid catalyst)
- Fries rearrangement
- Enantioselective glyoxalate-ene reactions
- Aldol reaction of silyl enol ethers with aldehydes.
- As an effective catalyst for electrophilic substitution reactions of indoles with imines.
- As catalyst for the synthesis of 4-aminocyclopentenones and functionalized azaspirocycles, via intramolecular aza-Piancatelli rearrangement.
- As new curing initiator to study the curing of diglycidyl ether of bisphenol-A (DGEBA).
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
The Fries rearrangement reaction is an organic name reaction which involves the conversion of phenolic esters into hydroxyaryl ketones on heating in the presence of a catalyst. Suitable catalysts for this reaction are Brønsted or Lewis acids such as HF, AlCl3, BF3, TiCl4, or SnCl4. The Fries rearrangement reaction is an ortho, para-selective reaction, and is used in the preparation of acyl phenols. This organic reaction has been named after German chemist Karl Theophil Fries.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico