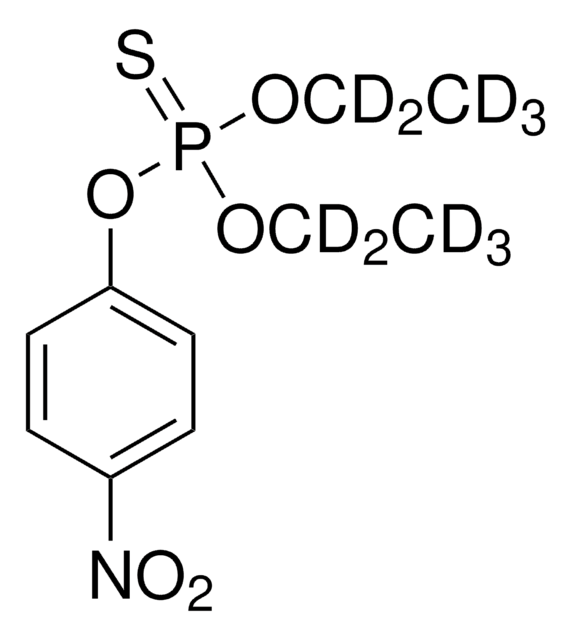
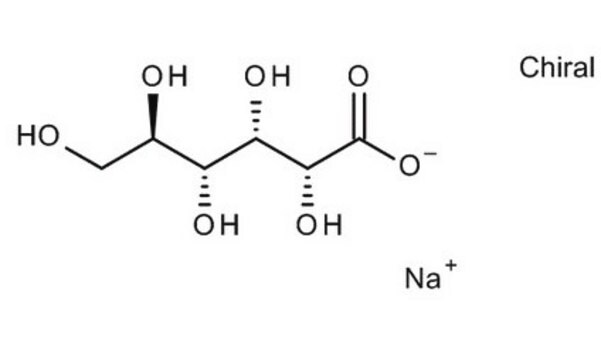
310301
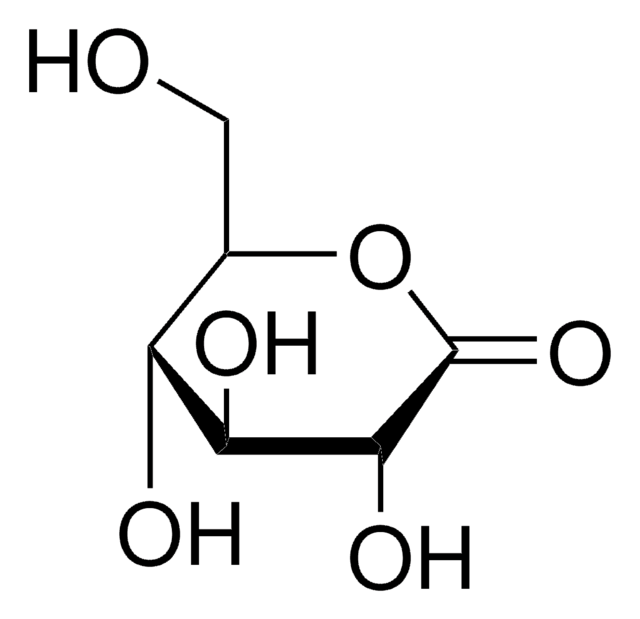
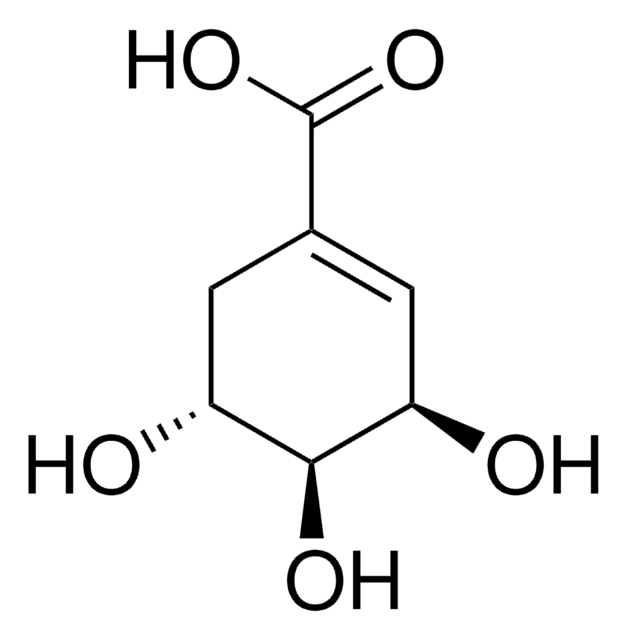
L-Gulonic acid γ-lactone
95%
Sinónimos:
L-Gulonic γ-lactone, L-(+)-Gulono-1,4-lactone
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C6H10O6
Número de CAS:
Peso molecular:
178.14
Beilstein/REAXYS Number:
83002
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
95%
form
solid
optical activity
[α]19/D +55°, c = 4 in H2O
mp
187-190 °C (lit.)
storage temp.
2-8°C
SMILES string
OC[C@H](O)[C@H]1OC(=O)[C@@H](O)[C@H]1O
InChI
1S/C6H10O6/c7-1-2(8)5-3(9)4(10)6(11)12-5/h2-5,7-10H,1H2/t2-,3+,4-,5+/m0/s1
InChI key
SXZYCXMUPBBULW-SKNVOMKLSA-N
Application
- Development and validation of an analysis method: Discusses the use of L-gulonic acid γ-lactone as a matrix effect inhibitor in the validation of a method for pesticide residues by gas chromatography–tandem mass spectrometry (Saegusa, Nomura, Takao, Hamaguchi, 2021).
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Beata A Wolucka et al.
The FEBS journal, 273(19), 4435-4445 (2006-09-08)
The last step of the biosynthesis of L-ascorbic acid (vitamin C) in plants and animals is catalyzed by L-gulono-1,4-lactone oxidoreductases, which use both L-gulono-1,4-lactone and L-galactono-1,4-lactone as substrates. L-gulono-1,4-lactone oxidase is missing in scurvy-prone, vitamin C-deficient animals, such as humans
Marjan Jeselnik et al.
Organic letters, 5(15), 2651-2653 (2003-07-19)
[reaction: see text] A new synthesis of L-noviose (11), a sugar moiety of novobiocin, is presented. D-Gulonolactone was initially converted in a few steps to the key ester derivative 7 [1-O-benzyl methyl 2,3-O-(1-methylethylidene)-alpha-L-lyxofuranosiduronate]. An appropriate selection of protecting groups enabled
G W Fleet et al.
Carbohydrate research, 205, 269-282 (1990-09-19)
The synthesis of the enantiomers of 6-epicastanospermine and of 1,6-diepicastanospermine from the enantiomeric gulonolactones is reported and the structure of the former is established as (1S,6R,7R,8R,8aR)-1,6,7,8-tetrahydroxyoctahydroindolizine. The inhibitory activities of the diastereomers against the amyloglucosidase-catalysed hydrolysis of p-nitrophenyl alpha-D-glucopyranoside were
F Puskás et al.
FEBS letters, 430(3), 293-296 (1998-08-04)
The orientation of gulonolactone oxidase activity was investigated in rat liver microsomes. Ascorbate formation upon gulonolactone addition resulted in higher intravesicular than extravesicular ascorbate concentrations in native microsomal vesicles. The intraluminal ascorbate accumulation could be prevented or the accumulated ascorbate
A Krasnov et al.
Genetic analysis : biomolecular engineering, 15(3-5), 115-119 (1999-12-22)
The reviewed studies addressed the possibility of using gene transfer for correction of L-ascorbic acid biosynthesis and carbohydrate utilization in rainbow trout. Analyses of enzymatic activities in the L-AAB pathway indicated that reasons for the lack of L-AA production can
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico