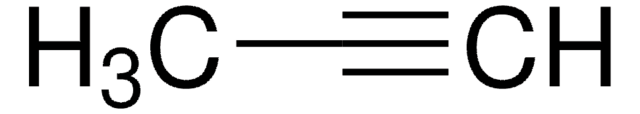256560
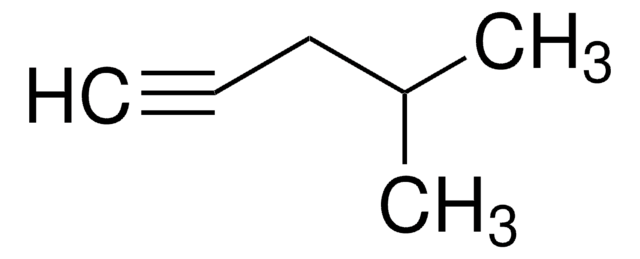
1-Pentyne
99%
Sinónimos:
Propylacetylene
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CH3CH2CH2C≡CH
Número de CAS:
Peso molecular:
68.12
Beilstein/REAXYS Number:
1697133
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
vapor pressure
6.8 psi ( 20 °C)
Quality Level
assay
99%
form
liquid
refractive index
n20/D 1.385 (lit.)
bp
40 °C (lit.)
mp
−106-−105 °C (lit.)
density
0.691 g/mL at 25 °C (lit.)
SMILES string
CCCC#C
InChI
1S/C5H8/c1-3-5-4-2/h1H,4-5H2,2H3
InChI key
IBXNCJKFFQIKKY-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Selective and non-selective hydrogenation of 1-pentyne catalyzed by silica-supported palladium has been studied by in situ X-ray absorption spectroscopy.
Application
1-Pentyne has been used in preparation of:
- lithium acetylides, required for asymmetric synthesis of α,α-dibranched propargyl sulfinamides
- 7-hydroxy-10-methoxy-3H-naphtho[2.1-b]pyrans
For use with
Referencia del producto
Descripción
Precios
signalword
Danger
hcodes
Hazard Classifications
Flam. Liq. 2
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
-4.0 °F - closed cup
flash_point_c
-20 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Manish Rawat et al.
Journal of the American Chemical Society, 128(34), 11044-11053 (2006-08-24)
The carbene complex 5-(2,2-dimethyl-2H-chromene)methoxylmethylene chromium pentacarbonyl will undergo a benzannulation reaction with phenylacetylene, 1-pentyne, 3-hexyne, and trimethylsilylacetylene to give 7-hydroxy-10-methoxy-3H-naphtho[2.1-b]pyrans as the primary product. These compounds are difficult to obtain pure due to their sensitivity to air. If the benzannulation
Andrew W Patterson et al.
The Journal of organic chemistry, 71(18), 7110-7112 (2006-08-26)
Addition of lithium acetylides prepared from 1-pentyne, phenylacetylene, and trimethylsilylacetylene to diverse N-tert-butanesulfinyl ketimines affords a range of alpha,alpha-dibranched propargyl sulfinamides in generally good yields (up to 87%) and with high diastereoselectivities (up to >99:1). Acidic cleavage of the tert-butanesulfinyl
Min Wei Tew et al.
Physical chemistry chemical physics : PCCP, 14(16), 5761-5768 (2012-03-17)
The catalytically active phase of silica-supported palladium catalysts in the selective and non-selective hydrogenation of 1-pentyne was determined using in situ X-ray absorption spectroscopy at the Pd K and L(3) edges. Upon exposure to alkyne, a palladium carbide-like phase rapidly
Quan-De Wang et al.
International journal of molecular sciences, 20(13) (2019-07-03)
Hydrogen atom abstraction from propargyl C-H sites of alkynes plays a critical role in determining the reactivity of alkyne molecules and understanding the formation of soot precursors. This work reports a systematic theoretical study on the reaction mechanisms and rate
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico