217735
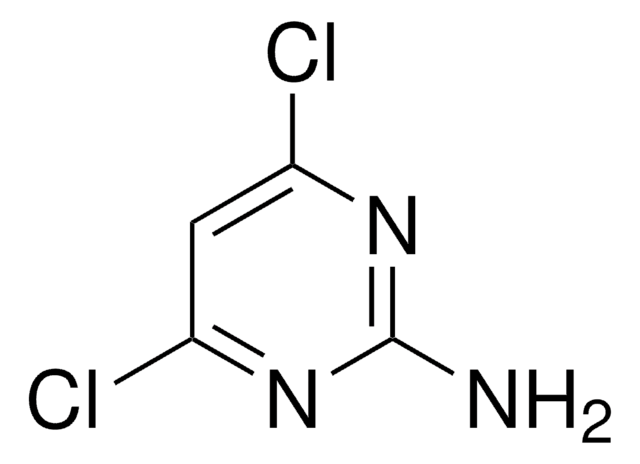
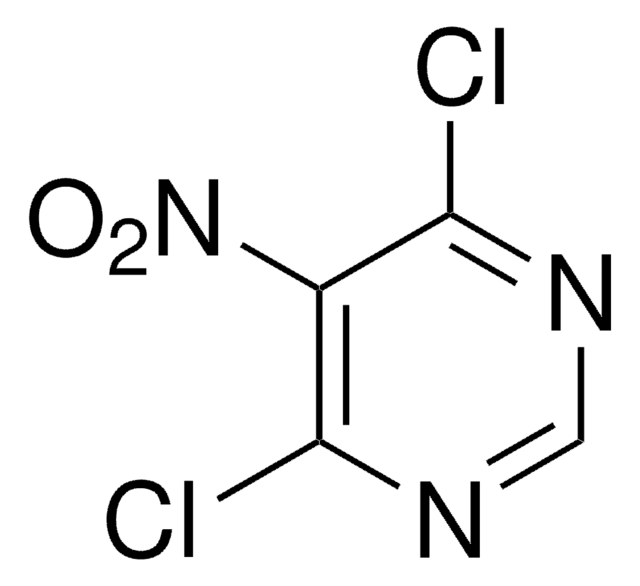
5-Amino-4,6-dichloropyrimidine
97%
About This Item
Productos recomendados
assay
97%
form
liquid
mp
145-148 °C (lit.)
solubility
95% ethanol: soluble 50 mg/mL, clear to slightly hazy, colorless to faintly yellow
functional group
chloro
storage temp.
2-8°C
SMILES string
Nc1c(Cl)ncnc1Cl
InChI
1S/C4H3Cl2N3/c5-3-2(7)4(6)9-1-8-3/h1H,7H2
InChI key
NIGDWBHWHVHOAD-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- oxepane ring containing monocyclic, conformationally restricted bicyclic and spirocyclic nucleosides
- conformationally locked bicyclo[2.2.1]heptane/oxa-bicyclo[3.2.1]octane nucleosides
- N(7)-substituted purines
- chiral derivatives of (+)-erythro-9-(2-hydroxy-3-nonyl)adenine
- 9-alkyl-6-substituted-purine analogs, potent anticonvulsant agents
- pyrimido-oxazepines in a three-step process with microwave heating at 150°C
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico