推薦產品
生物源
plant (Withania somnifera)
品質等級
化驗
≥95% (HPLC)
形狀
powder
mp
252-253 °C
官能基
epoxy
ketone
運輸包裝
ambient
儲存溫度
2-8°C
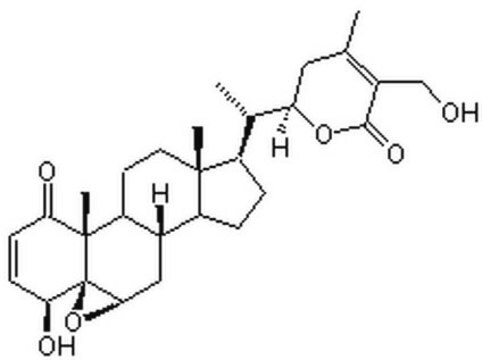
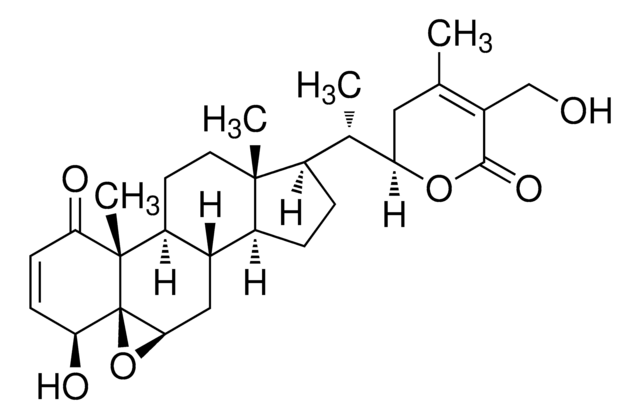
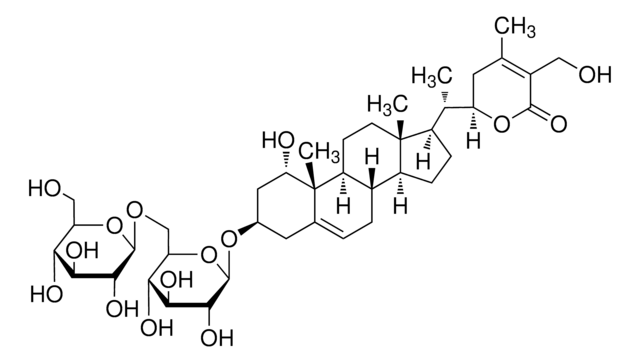
SMILES 字串
C[C@H]([C@H]1CC(C)=C(CO)C(=O)O1)[C@H]2CC[C@H]3[C@@H]4C[C@H]5O[C@]56[C@@H](O)C=CC(=O)[C@]6(C)[C@H]4CC[C@]23C
InChI
1S/C28H38O6/c1-14-11-21(33-25(32)17(14)13-29)15(2)18-5-6-19-16-12-24-28(34-24)23(31)8-7-22(30)27(28,4)20(16)9-10-26(18,19)3/h7-8,15-16,18-21,23-24,29,31H,5-6,9-13H2,1-4H3/t15-,16-,18+,19-,20-,21+,23-,24+,26+,27-,28+/m0/s1
InChI 密鑰
DBRXOUCRJQVYJQ-CKNDUULBSA-N
應用
用醉茄素 A 处理 HEK293 细胞,研究其对囊性纤维化炎症的影响。
生化/生理作用
对肿瘤细胞具有细胞毒性的甾体内酯。保护作用归因于抗脂质过氧化、抗氧化和解毒功能。
醉茄素 A 是从植物中分离得到的甾体内酯 。具有强大的抗炎属性,可抑制 NF-κ 的活化B 信号通路。醉茄素 A 的抗肿瘤活性是由于它能够通过结合膜联蛋白 II 和破坏 F 肌动蛋白交联来改变细胞骨架结构。醉茄素 A 通过与波形蛋白和肌动蛋白结合抑制血管生成。
準備報告
醉茄素 A 在 1 mg/mL 甲醇中生成澄清、无色溶液。
儲存類別代碼
13 - Non Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
438.8 °F
閃點(°C)
226 °C
客戶也查看了
Paola Bargagna-Mohan et al.
Chemistry & biology, 14(6), 623-634 (2007-06-23)
The natural product withaferin A (WFA) exhibits antitumor and antiangiogenesis activity in vivo, which results from this drug's potent growth inhibitory activities. Here, we show that WFA binds to the intermediate filament (IF) protein, vimentin, by covalently modifying its cysteine
Silvia D Stan et al.
Nutrition and cancer, 60 Suppl 1, 51-60 (2008-11-15)
Withaferin A (WA) is derived from the medicinal plant Withania somnifera that has been safely used for centuries in the Indian Ayurvedic medicine for treatment of various ailments. We now demonstrate that WA treatment causes G2 and mitotic arrest in
Inhibition of NFκB by the natural product Withaferin A in cellular models of Cystic Fibrosis inflammation
Maitra R et al
Journal of Inflammation (London, England), 6, doi: 10-doi: 10 (2009)
Kirti Vaishnavi et al.
PloS one, 7(9), e44419-e44419 (2012-09-14)
Withanolides are naturally occurring chemical compounds. They are secondary metabolites produced via oxidation of steroids and structurally consist of a steroid-backbone bound to a lactone or its derivatives. They are known to protect plants against herbivores and have medicinal value
Azariyas A Challa et al.
PloS one, 7(8), e42989-e42989 (2012-08-18)
Type I collagen is the most abundant protein in the human body. Its excessive synthesis results in fibrosis of various organs. Fibrosis is a major medical problem without an existing cure. Excessive synthesis of type I collagen in fibrosis is
條款
Separation of Withanoside IV; Withanoside V; Withaferin A; 12-Deoxywithastramonolide; Withanolide A; Withanolide B
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務