推薦產品
品質等級
化驗
≥98% (HPLC)
形狀
powder
顏色
orange
溶解度
DMSO: ≥10 mg/mL
起源
Eli Lilly
儲存溫度
2-8°C
SMILES 字串
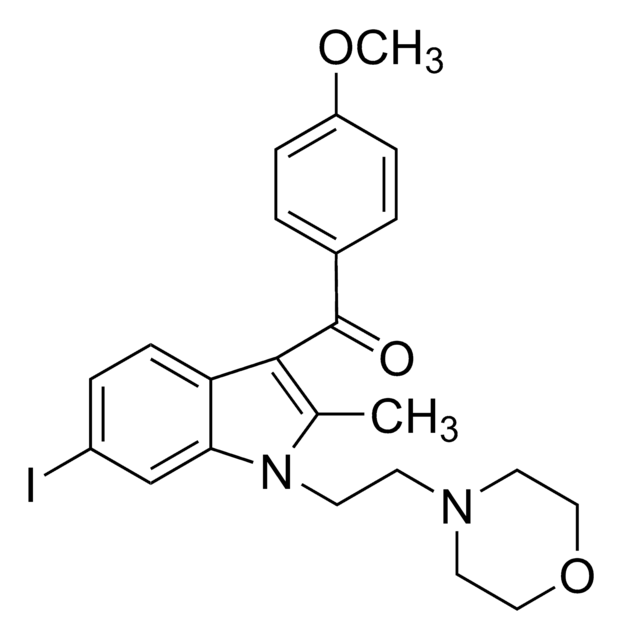
COc1ccc(cc1)-c2oc3cc(OC)ccc3c2C(=O)c4ccc(cc4)C#N
InChI
1S/C24H17NO4/c1-27-18-9-7-17(8-10-18)24-22(20-12-11-19(28-2)13-21(20)29-24)23(26)16-5-3-15(14-25)4-6-16/h3-13H,1-2H3
InChI 密鑰
RYNSGDFWBJWWSZ-UHFFFAOYSA-N
生化/生理作用
LY320135 is a potent CB1 receptor antagonist/inverse agonist (Ki = 141 nM) with greater than 70-fold selectivity over CB2 receptors (Ki > 10 μM).
LY320135 is a potent CB1 receptor antagonist/inverse agonist (Ki = 141 nM) with greater than 70-fold selectivity over CB2 receptors (Ki > 10 μM). Structurally dissimilar from SR 141716A and AM 251. Shows weak binding to both 5-HT2 (Ki = 6.4 μM) and muscarinic receptors (Ki = 2.1 μM)
特點和優勢
This compound is featured on the Cannabinoid Receptors page of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
This compound was developed by Eli Lilly. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 3 Oral
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
客戶也查看了
K M Kampa-Schittenhelm et al.
EBioMedicine, 54, 102678-102678 (2020-04-10)
Overriding the differentiation blockage in acute myeloid leukemia (AML) is the most successful mode-of-action in leukemia therapy - now curing the vast majority of patients with acute promyelocytic leukemia (APL) using all-trans retinoic acid (ATRA)-based regimens. Similar approaches in other
Xinwen Zhang et al.
Neuropharmacology, 128, 269-281 (2017-10-25)
Monoacylglycerol lipase (MGL) hydrolyzes 2-arachidonoylglycerol to arachidonic acid and glycerol. Inhibition of MGL may attenuate neuroinflammation by enhancing endocannabinoid signaling and decreasing prostaglandin (PG) production. Almost half of HIV infected individuals are afflicted with HIV-associated neurocognitive disorder (HAND), a neuroinflammatory
Elham Khajehali et al.
Molecular pharmacology, 88(2), 368-379 (2015-06-06)
CB1 cannabinoid receptors (CB1Rs) are attractive therapeutic targets for numerous central nervous system disorders. However, clinical application of cannabinoid ligands has been hampered owing to their adverse on-target effects. Ligand-biased signaling from, and allosteric modulation of, CB1Rs offer pharmacological approaches
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務