推薦產品
等級
analytical standard, for drug analysis
品質等級
drug control
Home Office Schedule 2; stupéfiant (France); kontrollierte Droge in Deutschland; (Spain); Decreto Lei 15/93: Tabela IIB (Portugal)
濃度
1.0 mg/mL in methanol
技術
HPLC: suitable
gas chromatography (GC): suitable
應用
pharmaceutical (small molecule)
格式
single component solution
儲存溫度
2-8°C
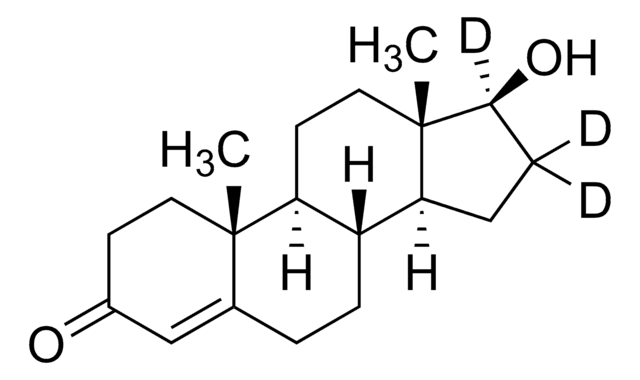
SMILES 字串
[H][C@@]12CCC(C)=C[C@@]1([H])c3c(O)cc(CCCCC)cc3OC2(C)C
InChI
1S/C21H30O2/c1-5-6-7-8-15-12-18(22)20-16-11-14(2)9-10-17(16)21(3,4)23-19(20)13-15/h11-13,16-17,22H,5-10H2,1-4H3/t16-,17-/m1/s1
InChI 密鑰
CYQFCXCEBYINGO-IAGOWNOFSA-N
基因資訊
human ... CNR1(1268) , CNR2(1269)
mouse ... Cnr1(12801)
rat ... Cnr1(25248)
尋找類似的產品? 前往 產品比較指南
一般說明
應用
- Development and validation of a high-performance liquid chromatographic method coupled with UV detection to measure cannabidiol and ∆9-tetrahydrocannabinol in rat plasma samples following their extraction by protein precipitation and liquid-liquid extraction (LLE)
- Estimation of cannabidiol and ∆9-tetrahydrocannabinol extracted from human plasma samples by liquid-liquid extraction using ultra-performance liquid chromatography with triple quadrupole mass spectrometry
- Multi-residue analysis of Δ9-tetrahydrocannabinol (THC) and its two degradation products, 11-hydroxy-Δ9-tetrahydrocannabinol and 11-nor-9-carboxy-Δ9-tetrahydrocannabinol, in whole blood samples by QuEChERS extraction and gas chromatography (GC) coupled with tandem mass spectrometry (MS/MS)
- Optimization of an ultra-high performance supercritical fluid chromatographic (UHPSFC) method, combined with a tandem mass spectrometer (MS/MS) to determine ∆9-tetrahydrocannabinol, along with three of its major metabolites and four synthetic metabolites in wastewater samples following liquid-liquid extraction
- Development and validation of an ultra high-performance liquid chromatographic (UHPLC) method coupled to photodiode array and mass spectrometry detectors to determine 11 cannabinoids in various Cannabis Sativa samples
其他說明
訊號詞
Danger
危險分類
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Flam. Liq. 2 - STOT SE 1
標靶器官
Eyes,Central nervous system
儲存類別代碼
3 - Flammable liquids
水污染物質分類(WGK)
WGK 2
閃點(°F)
49.5 °F - closed cup
閃點(°C)
9.7 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
客戶也查看了
文章
Tetrahydrocannabinolic acid A solution, 1.0 mg/mL in acetonitrile, ampule of 1 mL, certified reference material.
Tetrahydrocannabinolic acid A solution, 1.0 mg/mL in acetonitrile, ampule of 1 mL, certified reference material.
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務