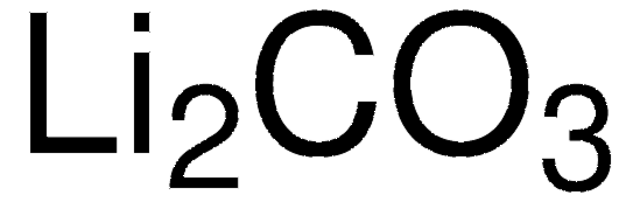推薦產品
品質等級
等級
battery grade
化驗
≥99.9% trace metals basis
形狀
powder
環保替代產品特色
Design for Energy Efficiency
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
雜質
≤1000 ppm (trace metals analysis)
mp
423 °C
溶解度
H2O: soluble ((lit.))
ethanol: slightly soluble ((lit.))
methanol: soluble ((lit.))
負離子痕跡
chloride (Cl-): ≤50 ppm
sulfate (SO42-): ≤50 ppm
應用
battery manufacturing
環保替代類別
SMILES 字串
[Li+].O.[OH-]
InChI
1S/Li.2H2O/h;2*1H2/q+1;;/p-1
InChI 密鑰
GLXDVVHUTZTUQK-UHFFFAOYSA-M
尋找類似的產品? 前往 產品比較指南
一般說明
Lithium hydroxide monohydrate is a white-to-colorless, crystalline salt. The monohydrate is hygroscopic. It is soluble in water and generates heat when dissolving. It is also soluble in methanol, somewhat soluble in ethanol, but only sparingly soluble in isopropanol.
Lithium hydroxide is produced in several ways. Most commonly, lithium carbonate is reacted with calcium hydroxide in a metathesis reaction. This directly yields lithium hydroxide hydrate, which is separated from the insoluble calcium carbonate byproduct and purified. Alternatively, when the source of lithium is spodumene ore, the ore can be converted to lithium hydroxide without first forming the carbonate. In the process, the lithium ore is treated with high-temperatures and sulfuric acid to form lithium sulfate; then the lithium sulfate is reacted with sodium hydroxide to form lithium hydroxide hydrate, which is purified.
Lithium hydroxide is produced in several ways. Most commonly, lithium carbonate is reacted with calcium hydroxide in a metathesis reaction. This directly yields lithium hydroxide hydrate, which is separated from the insoluble calcium carbonate byproduct and purified. Alternatively, when the source of lithium is spodumene ore, the ore can be converted to lithium hydroxide without first forming the carbonate. In the process, the lithium ore is treated with high-temperatures and sulfuric acid to form lithium sulfate; then the lithium sulfate is reacted with sodium hydroxide to form lithium hydroxide hydrate, which is purified.
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for energy efficiency. Click here for more information.
應用
The primary application of battery-grade lithium hydroxide is in the synthesis and manufacturing of cathode materials for lithium-ion batteries. In particular, lithium hydroxide is the reagent of choice for making nickel-rich cathodes like nickel-manganese-cobalt oxide (NMC) and nickel-cobalt-aluminum oxide (NCA). For these materials, the nickel-rich precursors must be fired in oxygen at relatively low temperatures (~500 °C) in order to promote higher oxidation states of nickel while suppressing cation mixing. Lithium hydroxide, which melts at 462 °C, is preferred because it melts at these temperatures, yielding more complete reactions and superior crystallinity, than reactions using lithium carbonate. Lithium carbonate, which melts at 723 °C, is still a solid at these temperatures.
Our battery grade lithium hydroxide monohydrate is well-suited for synthesis of nickel-rich metal oxides, like lithium nickel-manganese-aluminum oxide (NMA) and complex quaternary transition metal oxides like Zr-doped or Ti-doped nickel-manganese oxide.
Our lithium hydroxide monohydrate can also be used to synthesize lithium iron phosphates like LiFePO4 or lithium manganese oxides like Li2Mn2O4.
Our battery grade lithium hydroxide monohydrate is well-suited for synthesis of nickel-rich metal oxides, like lithium nickel-manganese-aluminum oxide (NMA) and complex quaternary transition metal oxides like Zr-doped or Ti-doped nickel-manganese oxide.
Our lithium hydroxide monohydrate can also be used to synthesize lithium iron phosphates like LiFePO4 or lithium manganese oxides like Li2Mn2O4.
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 1
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Chemical and Magnetic Characterization of Spinel Materials in the LiMn2O4?Li2Mn4O9?Li4Mn5O12 System.
Masquelie C, et al.
Journal of Solid State Chemistry, 123, 255-266 (1996)
A perspective on single-crystal layered oxide cathodes for lithium-ion batteries.
Langdon J, et al.
Energy Storage Materials, 37, 143-160 (2021)
Designing principle for Ni-rich cathode materials with high energy density for practical applications.
Xia Y, et al.
Nano Energy, 49, 434-452 (2018)
Li Wang et al.
Nano letters, 12(11), 5632-5636 (2012-10-19)
We report the crystal orientation tuning of LiFePO(4) nanoplates for high rate lithium battery cathode materials. Olivine LiFePO(4) nanoplates can be easily prepared by glycol-based solvothermal process, and the largest crystallographic facet of the LiFePO(4) nanoplates, as well as so-caused
Electrochemical and Structural Properties of xLi2M`O3?(1?x)LiMn0.5Ni0.5O2 Electrodes for Lithium Batteries (M` = Ti, Mn, Zr; 0 ? x ? 0.3).
Chemistry of Materials, 16, 1996-2006 (2004)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務