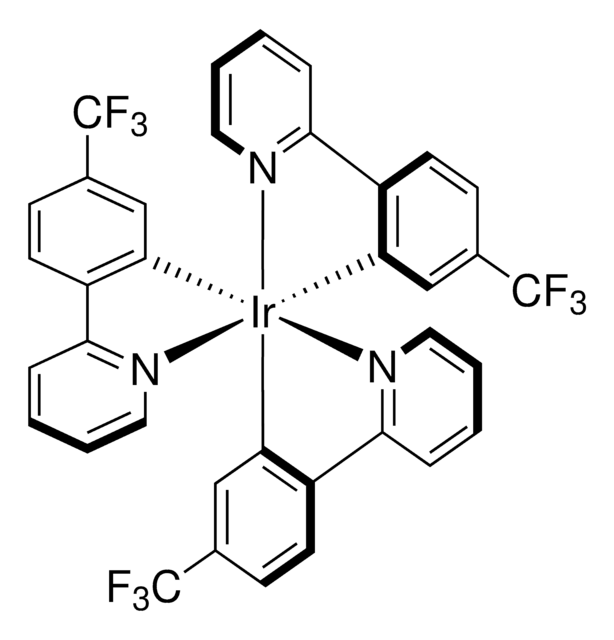
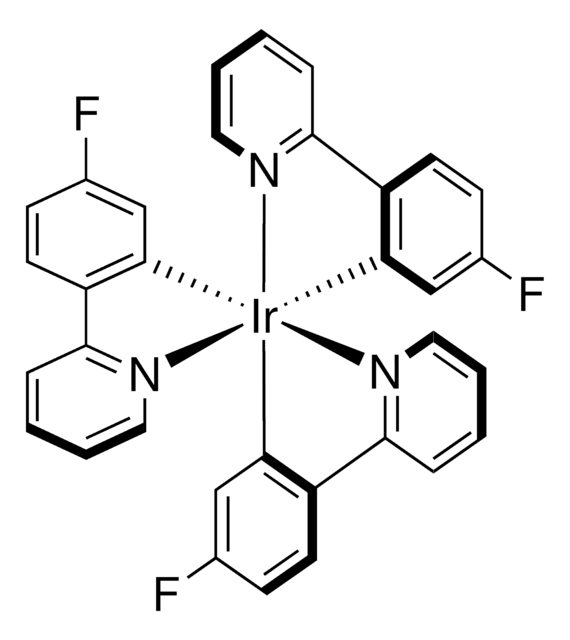
推薦產品
形狀
powder
反應適用性
reaction type: Photocatalysis
reagent type: catalyst
SMILES 字串
[Ir](c5c(ccc(c5)C(C)(C)C)c6ncccc6)(c3c(ccc(c3)C(C)(C)C)c4ncccc4)c1c(ccc(c1)C(C)(C)C)c2ncccc2
InChI
1S/3C15H16N.Ir/c3*1-15(2,3)13-9-7-12(8-10-13)14-6-4-5-11-16-14;/h3*4-7,9-11H,1-3H3;
InChI 密鑰
HVSYDKXCADBJCU-UHFFFAOYSA-N
應用
This photocatalyst facilitates a variety of transformations using visible light, including the benzylation of aldehydes with alcohols.
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
其他說明
Spin-Center Shift-Enabled Direct Enantioselective α-Benzylation of Aldehydes with Alcohols
Photocatalytic Generation of 2-Azolyl Radicals: Intermediates for the Azolylation of Arenes and Heteroarenes via C–H Functionalization
Visible-Light Photocatalytic Decarboxylation of α,β-Unsaturated Carboxylic Acids: Facile Access to Stereoselective Difluoromethylated Styrenes in Batch and Flow
Photocatalytic Generation of 2-Azolyl Radicals: Intermediates for the Azolylation of Arenes and Heteroarenes via C–H Functionalization
Visible-Light Photocatalytic Decarboxylation of α,β-Unsaturated Carboxylic Acids: Facile Access to Stereoselective Difluoromethylated Styrenes in Batch and Flow
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Xiao-Jing Wei et al.
ACS catalysis, 7(10), 7136-7140 (2017-11-08)
The development of synthetic methodologies which provide access to both stereoisomers of α,β-disubstituted olefins is a challenging undertaking. Herein, we describe the development of an operationally simple and stereoselective synthesis of difluoromethylated styrenes via a visible-light photocatalytic decarboxylation strategy using
Amandeep Arora et al.
Organic letters, 18(16), 3996-3999 (2016-08-06)
The 2-azolyl radical, generated from 2-bromoazoles via photocatalysis, is a powerful intermediate for the intermolecular arylation of unmodified (hetero)arenes. The reaction is characterized by mild conditions, operational simplicity, tolerance toward functional and sterically demanding groups, broad scope, and anti-Minisci selectivity.
Eric D Nacsa et al.
Journal of the American Chemical Society, 140(9), 3322-3330 (2018-02-06)
Nature routinely engages alcohols as leaving groups, as DNA biosynthesis relies on the removal of water from ribonucleoside diphosphates by a radical-mediated "spin-center shift" (SCS) mechanism. Alcohols, however, remain underused as alkylating agents in synthetic chemistry due to their low
Facile synthesis and complete characterization of homoleptic and heteroleptic cyclometalated Iridium(III) complexes for photocatalysis
Singh A, et al.
Journal of Organometallic Chemistry, 776, 51-59 (2015)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)
![三[2-苯基吡啶-C2,N]铱(III) 97%](/deepweb/assets/sigmaaldrich/product/structures/167/234/658d0b76-d31d-4fd5-8041-e04e207227c9/640/658d0b76-d31d-4fd5-8041-e04e207227c9.png)
![[Ir(dtbbpy)(ppy)2]PF6](/deepweb/assets/sigmaaldrich/product/structures/158/329/2544d673-d267-4aa1-8f46-2652aad4bfa0/640/2544d673-d267-4aa1-8f46-2652aad4bfa0.png)


![Ir[dF(t-Bu)-ppy]3](/deepweb/assets/sigmaaldrich/product/structures/254/294/d0fb19e5-05b2-4c1b-990b-a99fa60b3e73/640/d0fb19e5-05b2-4c1b-990b-a99fa60b3e73.png)
![[铱 (dF (Me) ppy)] 2 (dtbbpy) ] PF 6](/deepweb/assets/sigmaaldrich/product/structures/150/099/7c2dfa31-39f4-4cca-aee5-86d4a89fea78/640/7c2dfa31-39f4-4cca-aee5-86d4a89fea78.png)
