推薦產品
化驗
≥95% anhydrous basis
形狀
powder or crystals
反應適用性
core: nickel
reaction type: Cross Couplings
reagent type: catalyst
mp
107.9-132.4 °C (Decomp)
應用
2,6-Bis(N-pyrazolyl)pyridine nickel (II) dichloride ((bpp)NiCl2) is a Ni precatalyst that can be used in Negishi alkyl-alkyl cross-coupling, reductive cross-coupling of styrenyl aziridines, and dialkyl ether formation.
相關產品
產品號碼
描述
訂價
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
客戶也查看了
Brian P Woods et al.
Journal of the American Chemical Society, 139(16), 5688-5691 (2017-04-14)
A Ni-catalyzed reductive cross-coupling of styrenyl aziridines with aryl iodides is reported. This reaction proceeds by a stereoconvergent mechanism and is thus amenable to asymmetric catalysis using a chiral bioxazoline ligand for Ni. The process allows facile access to highly
Dialkyl Ether Formation by Nickel-Catalyzed Cross-Coupling of Acetals and Aryl Iodides.
Arendt KM and Doyle AG
Angewandte Chemie (International Edition in English), 54(34), 9876-9880 (2015)
Nickel-Catalyzed Negishi Cross-Couplings of Secondary Nucleophiles with Secondary Propargylic Electrophiles at Room Temperature.
Smith SW and Fu GC
Angewandte Chemie (International ed. in English), 47(48), 9334-9336 (2008)
Nickel-catalyzed enantioselective reductive cross-coupling of styrenyl aziridines.
Woods BP, et al.
Journal of the American Chemical Society, 139(16), 5688-5691 (2017)
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 904937-250MG |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務![[4,4′-双(1,1-二甲基乙基)-2,2′-联吡啶]二氯化镍(II)](/deepweb/assets/sigmaaldrich/product/structures/471/091/6faa29b1-bf8a-4d87-90b2-4cc55e082620/640/6faa29b1-bf8a-4d87-90b2-4cc55e082620.png)

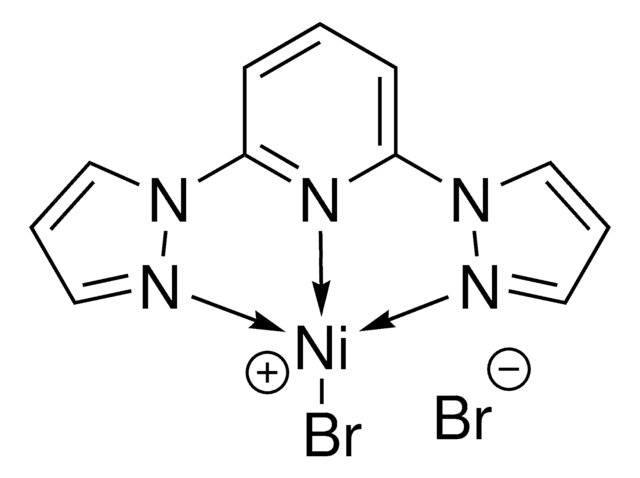

![[(TMEDA)Ni(o-甲苯基)Cl] 95%](/deepweb/assets/sigmaaldrich/product/structures/236/439/768c916e-994f-47e3-a980-3ca0471317d7/640/768c916e-994f-47e3-a980-3ca0471317d7.png)
![[1,3-双(二苯基膦)丙烷]二氯化镍(II)](/deepweb/assets/sigmaaldrich/product/structures/844/065/af07f787-c6a3-4a6e-a22b-47a933c73978/640/af07f787-c6a3-4a6e-a22b-47a933c73978.png)
![[1,1′-双(二苯基膦)二茂铁]二氯化镍(II) 97%](/deepweb/assets/sigmaaldrich/product/structures/274/566/a60d6584-163a-4c41-a738-60f8e4d524fa/640/a60d6584-163a-4c41-a738-60f8e4d524fa.png)