全部照片(1)
About This Item
經驗公式(希爾表示法):
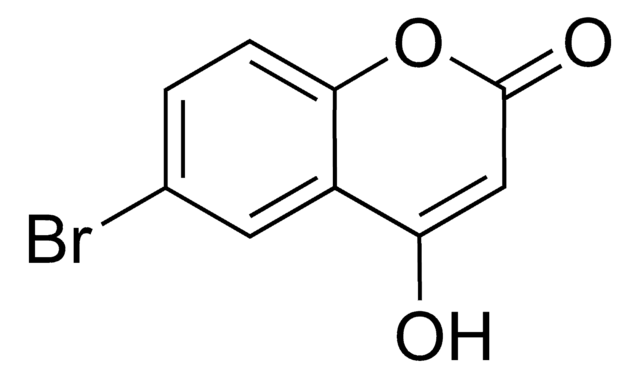
C9H5ClO3
CAS號碼:
分子量::
196.59
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推薦產品
品質等級
化驗
97%
mp
291 °C (dec.) (lit.)
官能基
chloro
ester
ketone
SMILES 字串
OC1=CC(=O)Oc2ccc(Cl)cc12
InChI
1S/C9H5ClO3/c10-5-1-2-8-6(3-5)7(11)4-9(12)13-8/h1-4,11H
InChI 密鑰
HUMZENGQNOATEQ-UHFFFAOYSA-N
一般說明
6-Chloro-4-hydroxycoumarin is a substituted 4-hydroxycoumarin. It participates in the Fe/Acetic acid (AcOH) catalyzed synthesis of various biologically active compounds.
應用
6-Chloro-4-hydroxycoumarin may be employed for the synthesis of 5-alkylidene-4-chloro-5H-1,2,3-dithiazoles. It may be employed for the synthesis of 6-[(coumarinyl-1-aryl-propionyl]benzoxazinones.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
客戶也查看了
Synthesis of 6-[1-(4'-hydroxycoumarin-3'-yl)-1-phenyl] propionyl-2H-[1,4]-benzoxazin-3(4H)-ones.
Jagath RG, et al.
Heterocyclic Communications, 10(1), 93-96 (2004)
Synthesis of new 5-alkylidene-4-chloro-5H-1, 2, 3-dithiazoles and their stereochemistry.
Jeon M-K and Kim K.
Tetrahedron, 55(32), 9651-9667 (1999)
Iron/acetic acid mediated intermolecular tandem C-C and C-N bond formation: an easy access to acridinone and quinoline derivatives.
Rajawinslin RR, et al.
Royal Society of Chemistry Advances, 4(71), 37806-37811 (2014)
Da-Wei Yan et al.
Molecules (Basel, Switzerland), 25(2) (2020-01-16)
Natural meroterpenes derived from phloroglucinols and β-caryophyllene have shown high inhibitory activity against α-glucosidase or cancer cells, however, the chemical diversity of this type of skeletons in Nature is limited. To expand the chemical space and explore their inhibitory activities
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務