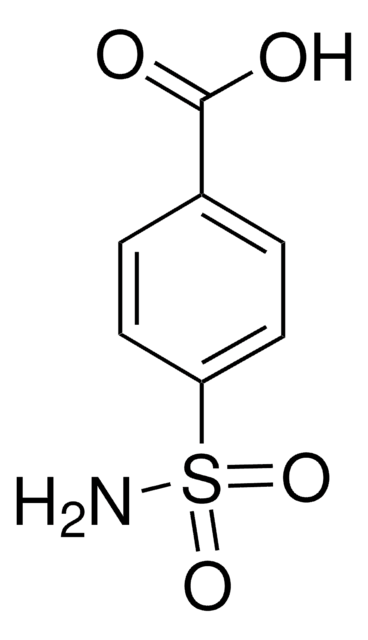
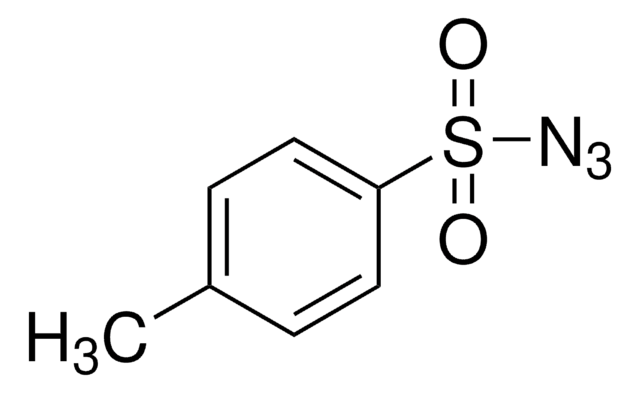
About This Item
推薦產品
品質等級
化驗
97%
反應適用性
reaction type: click chemistry
mp
180 °C (dec.) (lit.)
官能基
azide
carboxylic acid
儲存溫度
2-8°C
SMILES 字串
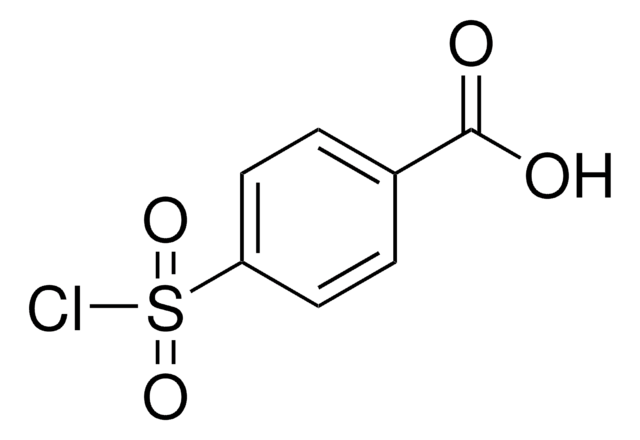
OC(=O)c1ccc(cc1)S(=O)(=O)N=[N+]=[N-]
InChI
1S/C7H5N3O4S/c8-9-10-15(13,14)6-3-1-5(2-4-6)7(11)12/h1-4H,(H,11,12)
InChI 密鑰
OWULJVXJAZBQLL-UHFFFAOYSA-N
應用
Synthesis of anti-inflammatory agents
Azide amidation
Reactions of thio acids with azides
Chemoselective sodium borohydride reduction of azides in water
Reagent for:
Photo-Stevens rearrangement
Cobalt-catalyzed synthesis of tertiary azides
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
客戶也查看了
文章
The chemistry of organoazides is exceedingly rich, since the azide functionality reacts with electrophiles, nucleophiles, and dipolarophiles, with or without the extrusion of dinitrogen. Common place transformation such as Staudinger reductions or ligations, Cu(I)-catalyzed Huisgen cycloadditions (of the “click” reaction family), Curtius or Schmidt rearrangents, nitrene reactions, or imine formation via aza-Wittig reactions all necessitate organoazide precursors or intermediates
The chemistry of organoazides is exceedingly rich, since the azide functionality reacts with electrophiles, nucleophiles, and dipolarophiles, with or without the extrusion of dinitrogen. Common place transformation such as Staudinger reductions or ligations, Cu(I)-catalyzed Huisgen cycloadditions (of the “click” reaction family), Curtius or Schmidt rearrangents, nitrene reactions, or imine formation via aza-Wittig reactions all necessitate organoazide precursors or intermediates
Organic Azides and Azide Sources
Global Trade Item Number
| 庫存單位 | GTIN |
|---|---|
| 340138-2.5G | 4061833554524 |
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務