推荐产品
分子量
~146,000 Da
包裝
pkg of 2 mg
製造商/商標名
USP
應用
pharmaceutical
形式
neat
儲存溫度
−70°C
一般說明
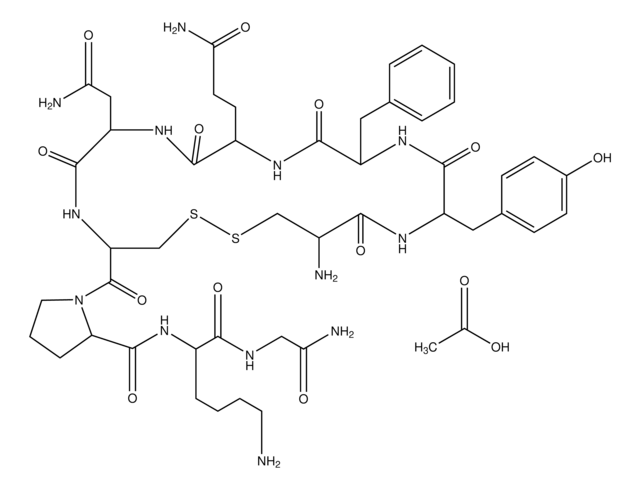
USP has developed four monoclonal antibody Reference Standards (mAb System Suitability, mAb 001, mAb 002, and mAb 003) to provide a range of reference materials with different physicochemical properties. These products underwent USP′s rigorous collaborative testing process including assessment by the USP General Chapter 129 methods.
Can be used as:
Can be used as:
- an independent control material for method development, training, and method transfer
- an internal assay control for standardization of physicochemical testing
應用
- Size-Exclusion Chromatography
- Capillary SDS Electrophoresis (Reduced and Nonreduced)
- Analysis of N-Linked Oligosaccharides
- Sialic Acid Analysis
- Intact Mass
- Sequence identification by peptide mapping
特點和優勢
- 4 Unique proteins with unique quality attributes
- Extensively characterized for confidence in method development
- Technical Note available
分析報告
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
其他說明
This product is part of the USP Biologics program.
Sales restrictions may apply.
儲存類別代碼
12 - Non Combustible Liquids
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门