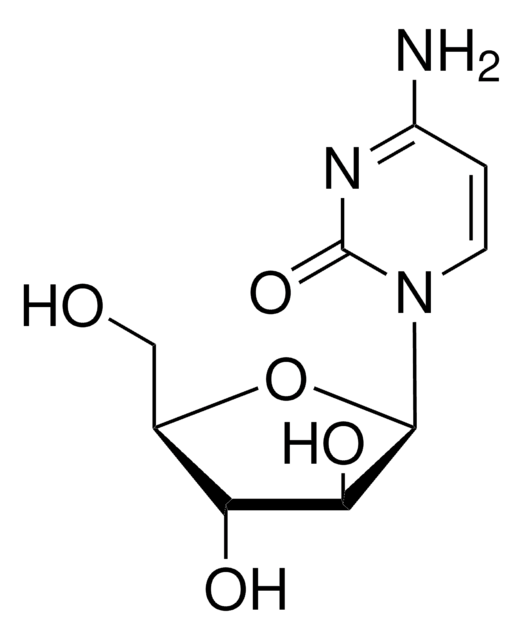
T3580
Toyocamycin
≥98% (HPLC), from Streptomyces rimosus
别名:
4-Aminopyrrolo[2,3-d]pyrimidine-5-carbonitrile 7-(β-D-ribofuranoside), 7-Deaza-7-cyanoadenosine, NSC 63701, NSC 99843, Neuro 000027, Unamycin B, Vengicide
About This Item
推荐产品
生物源
Streptomyces rimosus
品質等級
化驗
≥98% (HPLC)
形狀
solid
溶解度
DMSO: soluble 0.90-1.10 mg/mL, clear, colorless
H2O: moderately soluble
aqueous acid: moderately soluble
ethanol: moderately soluble
methanol: moderately soluble
抗生素活性譜
fungi
作用方式
DNA synthesis | interferes
儲存溫度
2-8°C
SMILES 字串
Nc1ncnc2n(cc(C#N)c12)[C@@H]3O[C@H](CO)[C@@H](O)[C@H]3O
InChI
1S/C12H13N5O4/c13-1-5-2-17(11-7(5)10(14)15-4-16-11)12-9(20)8(19)6(3-18)21-12/h2,4,6,8-9,12,18-20H,3H2,(H2,14,15,16)/t6-,8-,9-,12-/m1/s1
InChI 密鑰
XOKJUSAYZUAMGJ-WOUKDFQISA-N
正在寻找类似产品? 访问 产品对比指南
生化/生理作用
特點和優勢
準備報告
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
商品
We offers many products related to adenosine receptors for your research needs.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门