所有图片(1)
About This Item
经验公式(希尔记法):
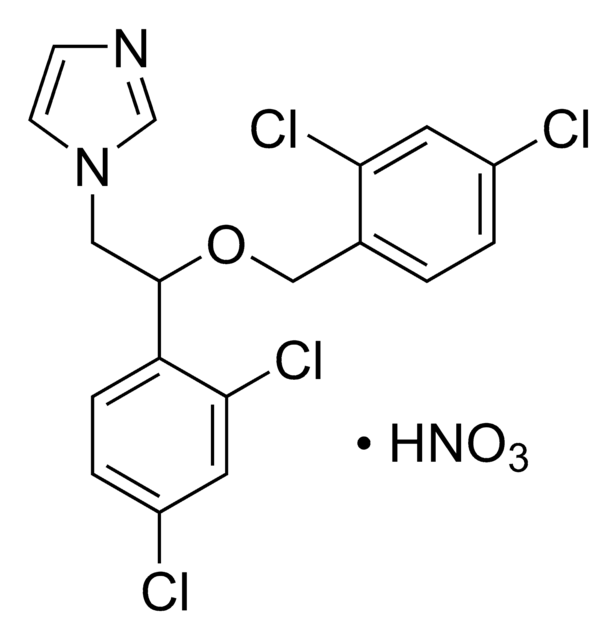
C18H15Cl3N2O · HNO3
CAS号:
分子量:
444.70
EC號碼:
MDL號碼:
分類程式碼代碼:
51102829
PubChem物質ID:
NACRES:
NA.85
推荐产品
形狀
powder or crystals
品質等級
顏色
white to off-white
抗生素活性譜
Gram-positive bacteria
fungi
作用方式
cell membrane | interferes
cell wall synthesis | interferes
enzyme | inhibits
SMILES 字串
O[N+]([O-])=O.Clc1ccc(COC(Cn2ccnc2)c3ccc(Cl)cc3Cl)cc1
InChI
1S/C18H15Cl3N2O.HNO3/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21;2-1(3)4/h1-9,12,18H,10-11H2;(H,2,3,4)
InChI 密鑰
DDXORDQKGIZAME-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
相关类别
一般說明
Chemical structure: imidazole
應用
Econazole is a broad spectrum antimycotic similar to ketoconazole. It has some action against Gram positive bacteria. It is used topically in dermatomycoses as well as orally and parenterally. It is used for studies on processes such as platelet serotonin uptake, prostanoid biosynthesis, EDHF-mediated relaxation, and Ca2+ transport pathways in thymic lymphocytes.
生化/生理作用
Econazole interacts with 14-α demethylase, a cytochrome P-450 enzyme necessary to convert lanosterol to ergosterol. Inhibition of ergosterol results in increased cellular permeability causing leakage of cellular contents. Econazole may also inhibit endogenous respiration, interact with membrane phospholipids, inhibit the transformation of yeasts to mycelial forms, inhibit purine uptake, and impair triglyceride and phospholipid biosynthesis.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
M A Ghannoum et al.
Clinical microbiology reviews, 12(4), 501-517 (1999-10-09)
The increased use of antibacterial and antifungal agents in recent years has resulted in the development of resistance to these drugs. The significant clinical implication of resistance has led to heightened interest in the study of antimicrobial resistance from different
Alessandro Malara et al.
The Journal of biological chemistry, 292(8), 3239-3251 (2017-01-05)
Abscisic acid (ABA) is a phytohormone involved in pivotal physiological functions in higher plants. Recently, ABA has been proven to be also secreted and active in mammals, where it stimulates the activity of innate immune cells, mesenchymal and hematopoietic stem
C J Jackson et al.
FEMS microbiology letters, 192(2), 159-162 (2000-11-07)
Azole antifungals are central to therapy and act by inhibiting a cytochrome P450, sterol 14-demethylase and blocking normal sterol synthesis. Our recent identification of a mycobacterial sterol biosynthetic pathway led us to probe the efficacy of a range of these
Inhibition of Ca2+ transport pathways in thymic lymphocytes by econazole, miconazole, and SKF 96365
M. J. Mason, B. Mayer, et al.
American Journal of Physiology. Cell Physiology, 264, C654-C662 (1993)
Núria Daranas et al.
Applied and environmental microbiology, 84(10) (2018-03-11)
A viability quantitative PCR (v-qPCR) assay was developed for the unambiguous detection and quantification of Lactobacillus plantarum PM411 viable cells in aerial plant surfaces. A 972-bp region of a PM411 predicted prophage with mosaic architecture enabled the identification of a
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门