推荐产品
等級
pharmaceutical primary standard
API 家族
dihydroergotamine
製造商/商標名
EDQM
應用
pharmaceutical (small molecule)
形式
neat
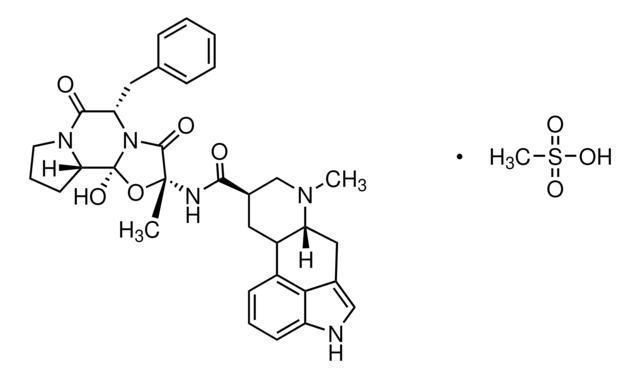
SMILES 字串
CS(O)(=O)=O.[H][C@@]12Cc3c[nH]c4cccc(C1C[C@H](CN2C)C(=O)N[C@]5(C)O[C@]6(O)N([C@@H](Cc7ccccc7)C(=O)N8CCC[C@@]68[H])C5=O)c34
InChI
1S/C33H37N5O5.CH4O3S/c1-32(35-29(39)21-15-23-22-10-6-11-24-28(22)20(17-34-24)16-25(23)36(2)18-21)31(41)38-26(14-19-8-4-3-5-9-19)30(40)37-13-7-12-27(37)33(38,42)43-32;1-5(2,3)4/h3-6,8-11,17,21,23,25-27,34,42H,7,12-16,18H2,1-2H3,(H,35,39);1H3,(H,2,3,4)/t21-,23?,25-,26+,27+,32-,33+;/m1./s1
InChI 密鑰
ADYPXRFPBQGGAH-WVVAGBSPSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
應用
Dihydroergotamine for peak identification EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
生化/生理作用
血管羟色胺受体激动剂;α-肾上腺素和多巴胺 D2 受体部分激动剂;血管收缩剂。
Vascular serotonin receptor agonist; vasoconstrictor.
包裝
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
其他說明
Sales restrictions may apply.
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Stephen Silberstein
Expert opinion on pharmacotherapy, 13(13), 1961-1968 (2012-08-07)
Dihydroergotamine mesylate (DHE) has been used as an acute migraine treatment since 1945, although tolerability with intravenous administration has limited its use. MAP0004 is a novel, orally inhaled, aerosol formulation of DHE that provides pulmonary drug delivery using a pressurized
John A Morren et al.
Expert opinion on pharmacotherapy, 11(18), 3085-3093 (2010-11-18)
Migraine affects approximately 18% of women and 6% of men, and has an immense impact on quality of life and productivity. Advancement in therapeutic options has been slow. For many patients with difficult-to-treat migraine, the appropriate use of dihydroergotamine mesylate
R B Lipton
Headache, 37 Suppl 1, S33-S41 (1997-01-01)
Ergotamine tartrate (ET) and dihydroergotamine mesylate (DHE) have been widely and effectively used in the treatment of migraine for many decades, although few randomized, controlled clinical trials have been conducted with these compounds. To compare their safety profiles, the world
J A Barone et al.
Pharmacotherapy, 6(4 Pt 2), 3S-11S (1986-07-01)
Dihydroergotamine(DHE)-heparin combination offers a unique treatment modality for the prevention of deep vein thrombosis. The combination appears to affect all 3 limbs of Virchow's triad: hypercoagulability, venous stasis, and endothelial damage. In most efficacy studies, data indicated that the combination
Drugs in R&D, 4(6), 376-377 (2003-10-31)
MT 300 is an injectable formulation of dihydroergotamine mesylate (DHE; a serotonin 5-HT1 receptor agonist) that is being developed by POZEN for the acute treatment of migraine. POZEN intends to present MT 300 as a prefilled syringe that can be
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门