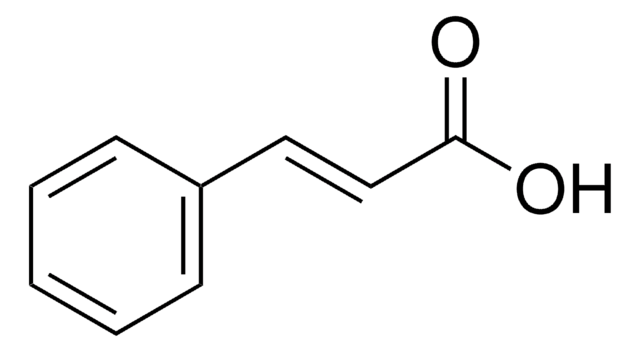
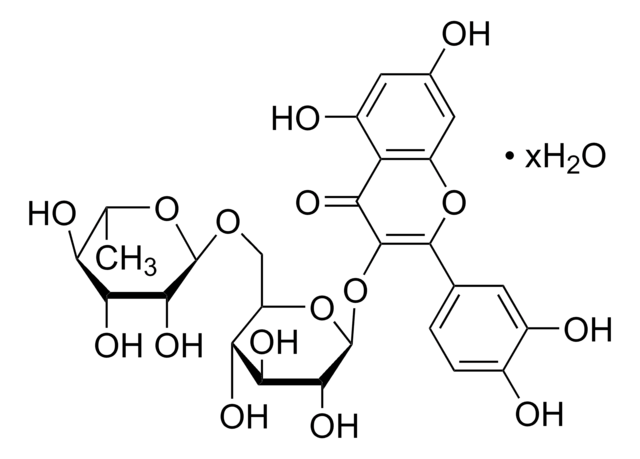
推荐产品
生物源
synthetic
品質等級
化驗
≥93% (H-NMR)
形狀
solid
技術
HPLC: suitable
顏色
dark red
mp
200—210 °C
儲存溫度
−20°C
SMILES 字串
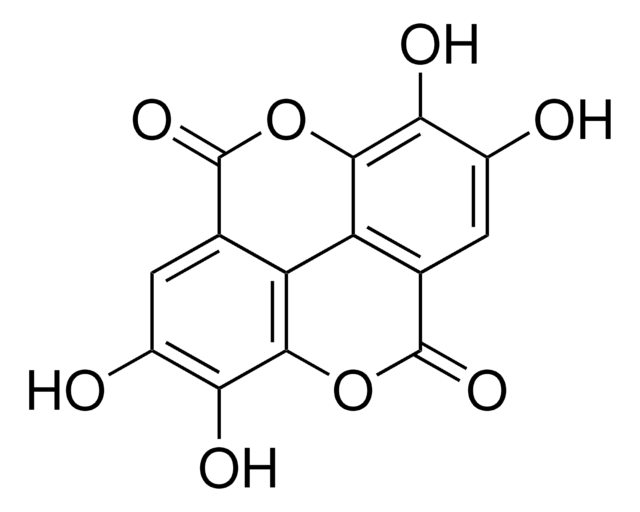
Canonical SMILES: CC1OCC2C(O1)C(C(C(O2)OC3C4COC(=O)C4C(C5=CC6=C(C=C35)OCO6)C7=CC(=O)C(=O)C(=C7)OC)O)OIsomeric SMILES: CC1OC[C@@H]2[C@@H](O1)[C@@H]([C@H]([C@@H](O2)OC3C4COC(=O)C4C(C5=CC6=C(C=C35)OCO6)C7=CC(=O)C(=O)C(=C7)OC)O)O
InChI
1S/C28H28O13/c1-10-35-8-19-26(39-10)23(31)24(32)28(40-19)41-25-13-6-17-16(37-9-38-17)5-12(13)20(21-14(25)7-36-27(21)33)11-3-15(29)22(30)18(4-11)34-2/h3-6,10,14,19-21,23-26,28,31-32H,7-9H2,1-2H3/t10?,14?,19-,20?,21?,23-,24-,25?,26-,28+/m1/s1
InChI 密鑰
SBLYXIKLMHGUJZ-LBMXFXLMSA-N
一般說明
應用
生化/生理作用
其他說明
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Carc. 1B - Repr. 2
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门