PHR1842
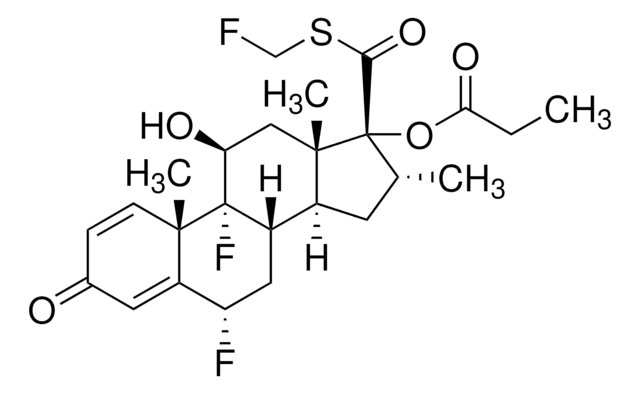
Fluticasone Related Compound A
Pharmaceutical Secondary Standard; Certified Reference Material
别名:
6ALPHA,9-DIFLUORO-11BETA-HYDROXY- 16ALPHA-METHYL-3-OXO-17 (PROPANOYLOXY)ANDROSTA-1,4-DIEN- 17BETA-CARBO(THIOPEROXOIC)SO-ACID (Fluticasone Related Compound A) (Fluticasone Impurity B), 6ALPHA,9-DIFLUORO-11BETA-HYDROXY- 16ALPHA-METHYL-3-OXO-17 (PROPANOYLOXY)ANDROSTA-1,4-DIEN- 17BETA-CARBO(THIOPEROXOIC)SO-ACID, ((6S,8S,9R,10S,11S,13S,14S,16R,17R)-6,9-difluoro-11-hydroxy-10,13,16-trimethyl-3-oxo-17-(propionyloxy)-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthrene-17-carbo(thioperoxoic)o-acid)
登录查看公司和协议定价
所有图片(1)
About This Item
推荐产品
一般說明
Fluticasone Related Compound B is an impurity of the synthetic corticosteroid drug, fluticasone propionate. Fluticasone propionate is generally used as an inhalation medication for the treatment of asthma.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
應用
Fluticasone Related Compound B may be used as a pharmaceutical reference standard for the determination of the analyte in bulk drug and pharmaceutical formulations by high performance liquid chromatography.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
分析報告
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
腳註
To see an example of a Certificate of Analysis for this material enter LRAB2754 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
相關產品
产品编号
说明
价格
訊號詞
Danger
危險聲明
危險分類
Resp. Sens. 1 - Skin Sens. 1
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Fluticasone propionate
USP42/NF37: United States Pharmacopeia and National Formulary
United States Pharmacopeia/National Formulary, 32(2), 1940-1940 (2014)
Comparative evaluation of the leukotriene receptor antagonist pranlukast versus the steroid inhalant fluticasone in the therapy of aged patients with mild bronchial asthma
Horiguchi T, et al.
Arzneimittelforschung, 57(02), 87-91 (2007)
Method Development and Validation of Simultaneous Determination of Related Substances for Salmeterol Xinafoate and Fluticasone Propionate in Dry Powder Inhalers
Arzu C, et al.
Chromatography, 10(02), 87-91 (2017)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门