推荐产品
品質等級
化驗
≥95%
形狀
solid
顏色
white to beige
儲存溫度
2-8°C
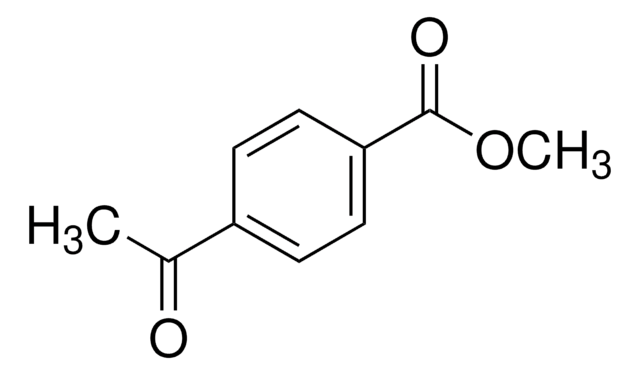
SMILES 字串
[N-]=[N+]=NCCCNC(=O)CCCCC1SCC2NC(=O)NC12
InChI
InChI=1S/C13H22N6O2S/c14-19-16-7-3-6-15-11(20)5-2-1-4-10-12-9(8-22-10)17-13(21)18-12/h9-10,12H,1-8H2,(H,15,20)(H2,17,18,21)/t9-,10-,12-/m0/s1
應用
This reagent enables the specific labeling of various alkynylated molecules, such as DNA, oligonucleotides, and proteins, with biotin. The binding of biotin to avidin or streptavidin can be employed in downstream affinity applications, such as the isolation of biotinylated molecules or their interaction with streptavidin conjugates. Biotin azide undergoes a copper-catalyzed click reaction with terminal alkynes, enabling the incorporation of biotin and biotin derivatives into biomolecules that contain alkyne groups through azide-alkyne cycloaddition.
特點和優勢
Biotin-azide (N-(3-Azidopropyl)biotinamide) is an azido derived biotin probe. Biotin-azide can be used to prepare various biotinylated conjugates via Click Chemistry.The conjugation of biotin and its derivatives to various biomolecules can be achieved through the widely recognized click chemistry methodology, followed by their detection using streptavidin, avidin, or NeutrAvidin biotin-binding proteins. Biotin azide serves as a valuable reagent for the synthesis of diverse biotinylated conjugates via Click Chemistry
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持
![N-[2-[2-[2-(2-叠氮乙氧基)乙氧基]乙氧基]乙基]生物素胺](/deepweb/assets/sigmaaldrich/product/structures/120/306/c9779b03-3754-4ad6-8eef-b07209e113ce/640/c9779b03-3754-4ad6-8eef-b07209e113ce.png)

![三[(1-苄基-1H-1,2,3-三唑-4-基)甲基]胺 97%](/deepweb/assets/sigmaaldrich/product/structures/179/695/86a721c8-2a4c-4e4f-bc36-6276ce7a941f/640/86a721c8-2a4c-4e4f-bc36-6276ce7a941f.png)