推荐产品
品質等級
化驗
≥95%
形狀
powder or solid
反應適用性
reagent type: catalyst
reaction type: Asymmetric synthesis
環保替代產品特色
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
官能基
amine
hydroxyl
imine
phenyl
環保替代類別
, Aligned
一般說明
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for catalytic efficiency. Click here for more information.
應用
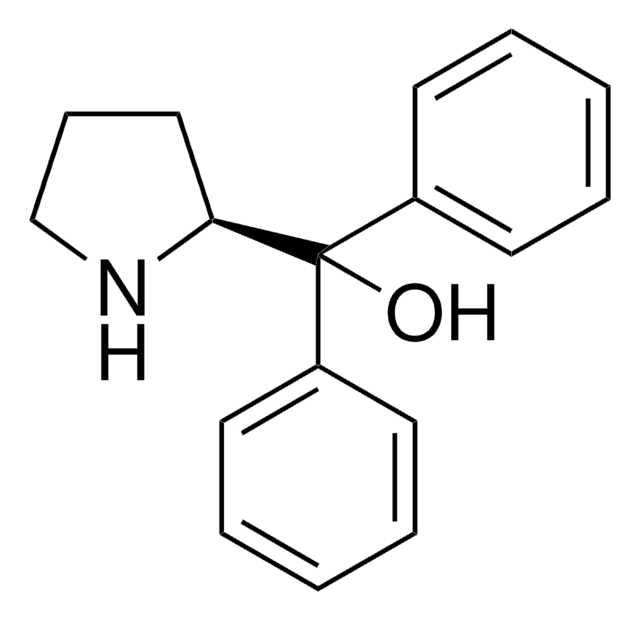
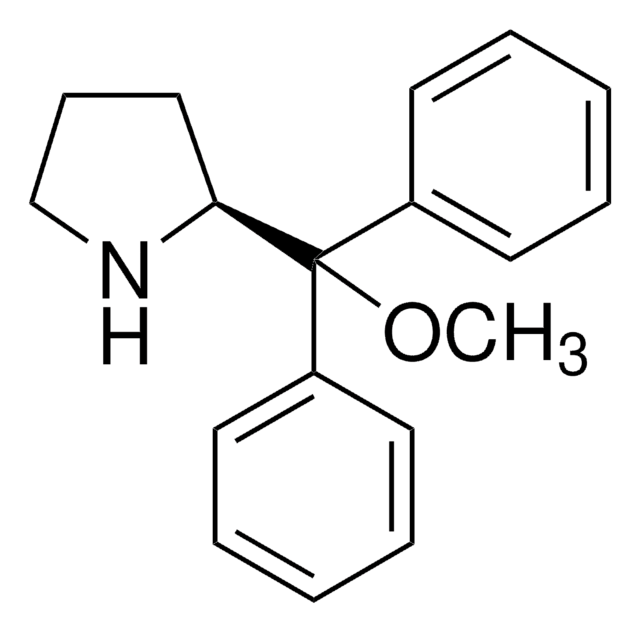
Chiral cyclopropenimines are a highly effective new class of enantioselective Brønsted base catalysts - the so-called “superbases” for enantioselective organocatalysis. Due to the prevalence of chemical reactions involving proton transfer as a key mechanistic event, Brønsted bases have become indispensable tools for the practice of organic synthetic chemistry, capable of catalyzing proton transfer reactions enantioselectively for the production of optically enriched products. Catalyst is stored as co-salt for stability. Conversion of the HCl salt to free catalyst requires a simple wash with aqueous base. This is one of a suite of Brønsted catalysts reported by Tristan Lambert and coworkers available through Sigma-Aldrich.
其他說明
Enantioselective Bronsted Base Catalysis with Chiral Cyclopropenimines
Cyclopropenimine-Catalyzed Enantioselective Mannich Reactions of tert-Butyl Glycinates with N-Boc-Imines
Transition State Analysis of Enantioselective Bronsted Base Catalysis by Chiral Cyclopropenimines
Structure-activity relationship studies of cyclopropenimines as enantioselective Bronsted base catalysts
Asymmetric Bronsted Base-Catalyzed and -Directed [3+2] Cycloaddition of 2-Acyl Cycloheptatrienes with Azomethine Ylides
Cyclopropenimine-Catalyzed Enantioselective Mannich Reactions of tert-Butyl Glycinates with N-Boc-Imines
Transition State Analysis of Enantioselective Bronsted Base Catalysis by Chiral Cyclopropenimines
Structure-activity relationship studies of cyclopropenimines as enantioselective Bronsted base catalysts
Asymmetric Bronsted Base-Catalyzed and -Directed [3+2] Cycloaddition of 2-Acyl Cycloheptatrienes with Azomethine Ylides
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Jeffrey S Bandar et al.
Journal of the American Chemical Society, 136(30), 10700-10707 (2014-07-17)
Experimental (13)C kinetic isotope effects have been used to interrogate the rate-limiting step of the Michael addition of glycinate imines to benzyl acrylate catalyzed by a chiral 2,3-bis(dicyclohexylamino) cyclopropenimine catalyst. The reaction is found to proceed via rate-limiting carbon-carbon bond
Jeffrey S Bandar et al.
Journal of the American Chemical Society, 135(32), 11799-11802 (2013-08-03)
Cyclopropenimine 1 is shown to catalyze Mannich reactions between glycine imines and N-Boc-aldimines with high levels of enantio- and diastereocontrol. The reactivity of 1 is shown to be substantially greater than that of a widely used thiourea cinchona alkaloid-derived catalyst.
Jeffrey S Bandar et al.
Chemical science, 6(2), 1537-1547 (2015-10-28)
We recently demonstrated that chiral cyclopropenimines are viable Brønsted base catalysts in enantioselective Michael and Mannich reactions. Herein, we describe a series of structure-activity relationship studies that provide an enhanced understanding of the effectiveness of certain cyclopropenimines as enantioselective Brønsted
Vibeke H Lauridsen et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 22(10), 3259-3263 (2016-01-26)
Conjugated cyclic trienes have the potential for different types of cycloaddition reactions. In the present work, we will, in a novel asymmetric cycloaddition reaction, demonstrate that the organocatalytic reaction of 2-acyl cycloheptatrienes with azomethine ylides proceeds as a [3+2] cycloaddition
Jeffrey S Bandar et al.
Journal of the American Chemical Society, 134(12), 5552-5555 (2012-03-16)
Cyclopropenimines are shown to be a highly effective new class of enantioselective Brønsted base catalysts. A chiral 2,3-bis(dialkylamino)cyclopropenimine catalyzes the rapid Michael reaction of a glycine imine substrate with high levels of enantioselectivity. A preparative scale reaction to deliver 25
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门