推荐产品
品質等級
化驗
97%
反應適用性
reaction type: Cross Couplings
reagent type: ligand
reaction type: Acylations
reagent type: ligand
reaction type: Arylations
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: C-C Bond Formation
reagent type: ligand
reaction type: Decarboxylations
reagent type: ligand
reaction type: Stille Coupling
mp
283-286 °C (lit.)
官能基
phosphine
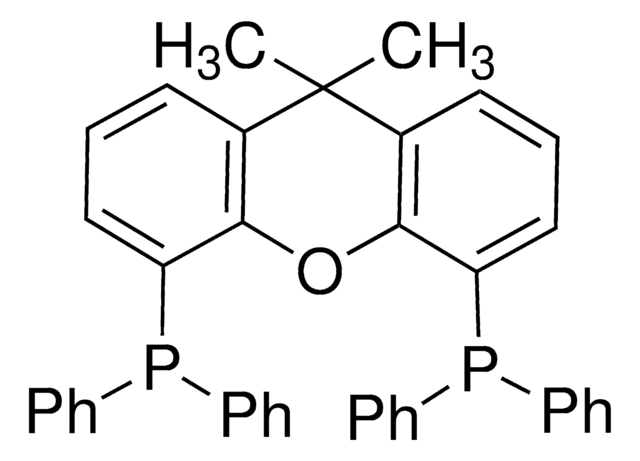
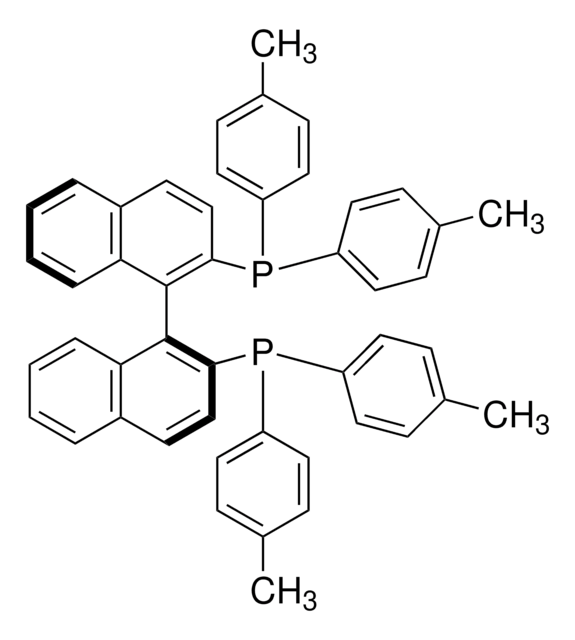
SMILES 字串
P(c8ccccc8)(c7ccccc7)c1c(c6c(cc1)cccc6)c2c3c(ccc2P(c5ccccc5)c4ccccc4)cccc3
InChI
1S/C44H32P2/c1-5-19-35(20-6-1)45(36-21-7-2-8-22-36)41-31-29-33-17-13-15-27-39(33)43(41)44-40-28-16-14-18-34(40)30-32-42(44)46(37-23-9-3-10-24-37)38-25-11-4-12-26-38/h1-32H
InChI 密鑰
MUALRAIOVNYAIW-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
應用
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
Global Trade Item Number
| 货号 | GTIN |
|---|---|
| 481084-100G | 4061833553305 |
| 481084-25G | 4061832388519 |
| 481084-500G | 4061833502709 |
| 481084-5G | 4061832388526 |
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持