所有图片(2)
About This Item
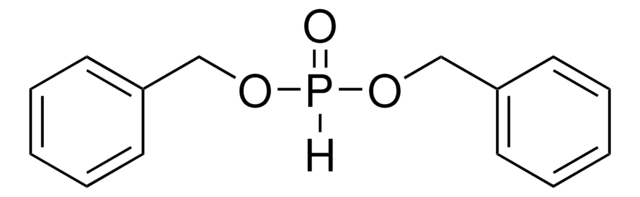
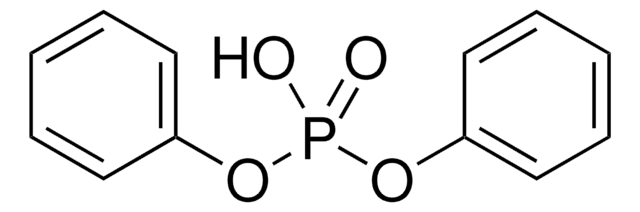
线性分子式:
(C6H5CH2O)2P(O)OP(O)(OCH2C6H5)2
CAS号:
分子量:
538.47
Beilstein:
2068292
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
品質等級
化驗
98%
mp
63-66 °C (lit.)
官能基
phenyl
phosphate
儲存溫度
−20°C
SMILES 字串
O=P(OCc1ccccc1)(OCc2ccccc2)OP(=O)(OCc3ccccc3)OCc4ccccc4
InChI
1S/C28H28O7P2/c29-36(31-21-25-13-5-1-6-14-25,32-22-26-15-7-2-8-16-26)35-37(30,33-23-27-17-9-3-10-18-27)34-24-28-19-11-4-12-20-28/h1-20H,21-24H2
InChI 密鑰
NSBNXCZCLRBQTA-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
相关类别
應用
焦磷酸四苄基可用于以下研究:
- Pb2+-选择性膜电极的制造。
- 制备合成核苷酸,3,6-二脱氧己糖的磷酸酯。
- 作为Und-PP-Bac(十一碳二烯基焦磷酸 = Und-PP;Bac =不寻常的糖杆菌胺)合成的磷酸化试剂。
訊號詞
Danger
危險聲明
危險分類
Skin Corr. 1B
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
其他客户在看
Solvolysis of Tetrabenzyl Pyrophosphate. Catalysis by Imidazole1
Blakeley R, et al.
Journal of the American Chemical Society, 88(1), 112-119 (1966)
Journal of the Chemical Society. Perkin Transactions 1, 729-729 (1992)
D Xu et al.
Talanta, 51(2), 365-371 (2008-10-31)
Tetrabenzyl pyrophosphate and diphenylphosphinic anhydride, with two phosphoryl groups (PO) as ligating sites, can be used as novel ionophores to make Pb(2+)-selective membrane electrodes. A good result was obtained with tetrabenzyl pyrophosphate, and the electrode based on this ionophore and
N S Utkina et al.
Bioorganicheskaia khimiia, 15(10), 1375-1383 (1989-10-01)
Interaction of lithium alcoholates of 2,4-di-O-benzoates of paratose and abequose with tetrabenzyl pyrophosphate gave alpha-phosphates of the 3,6-dideoxyhexoses, further converted into the corresponding cytidine-5'-diphosphate derivatives. These synthetic nucleotides were shown to participate in the biosynthesis of the O-specific polysaccharides for
Eranthie Weerapana et al.
Journal of the American Chemical Society, 127(40), 13766-13767 (2005-10-06)
The chemical synthesis and biological activity of undecaprenyl pyrophosphate bacillosamine (Und-PP-Bac), an obligatory intermediate in the asparagine-linked glycosylation pathway of Campylobacter jejuni, are reported. The key transformation involves the coupling of bacillosamine phosphate and undecaprenyl phosphate. The synthetic Und-PP-Bac can
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门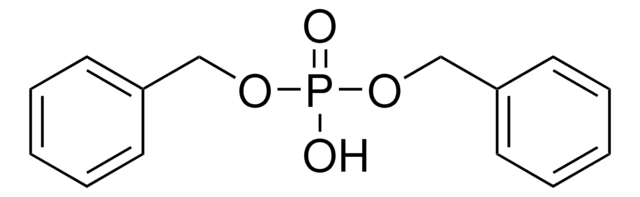



![1,8-二氮杂双环[5.4.0]十一碳-7-烯 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)