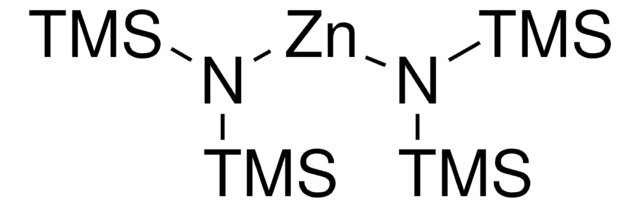推荐产品
化驗
99%
形狀
solid
光學活性
[α]20/D +280°, c = 1 in ethyl acetate
mp
200 °C (dec.) (lit.)
官能基
ether
hydroxyl
ketal
SMILES 字串
CC1(C)O[C@@H]([C@H](O1)C(O)(c2cccc3ccccc23)c4cccc5ccccc45)C(O)(c6cccc7ccccc67)c8cccc9ccccc89
InChI
1S/C47H38O4/c1-45(2)50-43(46(48,39-27-11-19-31-15-3-7-23-35(31)39)40-28-12-20-32-16-4-8-24-36(32)40)44(51-45)47(49,41-29-13-21-33-17-5-9-25-37(33)41)42-30-14-22-34-18-6-10-26-38(34)42/h3-30,43-44,48-49H,1-2H3/t43-,44-/m0/s1
InChI 密鑰
WTZVNZRNIOJACO-CXNSMIOJSA-N
應用
- For the highly enantioselective addition of primary alkyl Grignards to ketones.
- As an organocatalyst for the activation of carbonyl functionality in vinylogous addition reaction of an aldehyde.
- As a chiral dopant in the preparation of cholesteric liquid crystal (CLC) having an aggregation-induced-emission dye.
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
商品
The chiral auxiliaries TADDOLs (α,α,α,α-tetraaryl-1,3-dioxolane-4,5- dimethanols) developed by Seebach's group have found numerous applications in asymmetric synthesis ranging from utilization as stoichiometric chiral reagents or in Lewis acid mediated reactions, to roles in catalytic hydrogenation and stereoregular metathesis polymerization.
Apart from numerous examples using TADDOLs in metal-catalyzed asymmetric reactions, Rawal recently reported that TADDOLs could be used as Brønsted acid organocatalysts in highly stereoselective hetero-Diels–Alder reactions.
Apart from numerous examples using TADDOLs in metal-catalyzed asymmetric reactions, Rawal recently reported that TADDOLs could be used as Brønsted acid organocatalysts in highly stereoselective hetero-Diels–Alder reactions.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门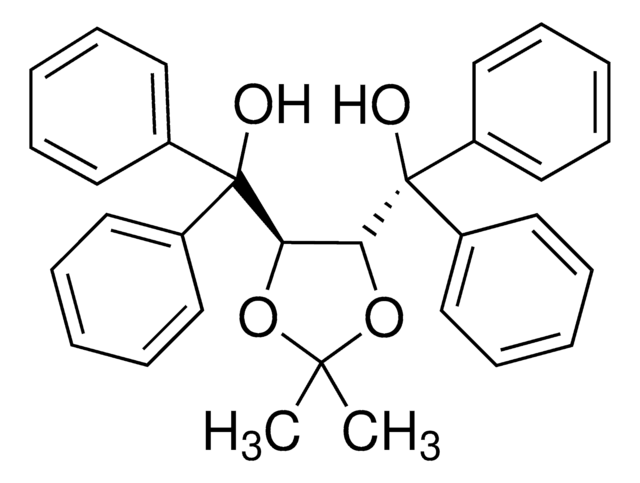
![氯环戊二烯基[(4R,5R)-2,2-二甲基-α,α,α′,α′-四苯基-1,3-二氧戊环-4,5-二甲醇酸根]钛 97%](/deepweb/assets/sigmaaldrich/product/structures/232/672/2ca86719-0965-4619-b25b-aae4087a2aad/640/2ca86719-0965-4619-b25b-aae4087a2aad.png)