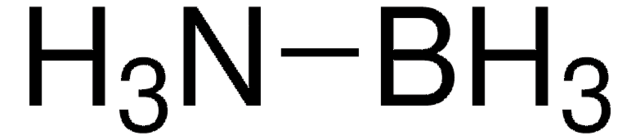推荐产品
蒸汽壓力
0.15 mmHg ( 20 °C)
品質等級
形狀
solid
反應適用性
reagent type: reductant
顏色
white
bp
213 °C (lit.)
mp
98-100 °C (lit.)
密度
0.94 g/mL at 25 °C (lit.)
正在寻找类似产品? 访问 产品对比指南
應用
Reactant for:
- Chemical vapor deposition of boron nitride nanosheets on Ni or Cu foils in the presence of ammonia
- Stereoselective catalytic hydrogenation reactions
- Chemical hydrogen storage
- Preparation of carborane-conjugated quinoline carboxamide ligands of translocator protein for boron neutron capture therapy (BNCT)
- Synthesis of hydrogenated boron clusters B12Hn with controlled hydrogen content
- Preparation of alkenyldecaboranes by regioselective transition-metal-catalyzed decaborane-alkyne hydroboration reactions
用于离子注入工艺的硼源
訊號詞
Danger
危險分類
Acute Tox. 2 Inhalation - Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Eye Irrit. 2 - Flam. Sol. 1 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
4.1B - Flammable solid hazardous materials
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges, type P3 (EN 143) respirator cartridges
Kiminori Ohta et al.
Bioorganic & medicinal chemistry letters, 14(23), 5913-5918 (2004-10-27)
We designed and synthesized novel retinoid X receptor (RXR)-selective antagonists bearing a carborane moiety. Compounds 8a-d or 9a-d themselves have no differentiation-inducing activity toward HL-60 cells and no inhibitory activity towards a retinoic acid receptor (RAR) agonist. However, they inhibit
Zhu Yinghuai et al.
Journal of the American Chemical Society, 129(20), 6507-6512 (2007-05-03)
Well dispersed ruthenium(0) nanoparticles, stabilized in the ionic liquid agent, trihexyltetradecylphosphonium dodecylbenzenesulfonate, have been successfully prepared via a reduction reaction of the precursor [CpRuCp*RuCp*]PF6 (Cp* = C5Me5). The ruthenium(0) nanoparticles were shown to catalyze the isotope exchange reaction between 10B
Ilias Sioutis et al.
The journal of physical chemistry. A, 110(45), 12528-12534 (2006-11-10)
The interaction of decaborane (B(10)H(14)) with the I(-) ion and the (isoelectronic) Xe atom is investigated using a number of theoretical methods: MP2, CCSD(T), CCSD, spin-orbit CISD, and DFT using the B3LYP, B3PW91, PW91PW91, and PBE0 methods. All non-DFT and
Yuqi Li et al.
Inorganic chemistry, 47(20), 9193-9202 (2008-09-20)
Unlike in conventional organic solvents, where Lewis base catalysts are required, decaborane dehydrogenative alkyne-insertion reactions proceed rapidly in biphasic ionic-liquid/toluene mixtures with a wide variety of terminal and internal alkynes, thus providing efficient, one-step routes to functional o-carborane 1-R-1,2-C2B10H11 and
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门