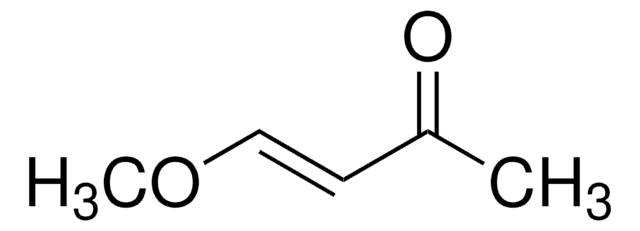
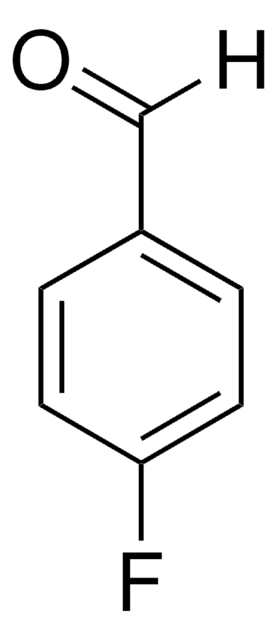
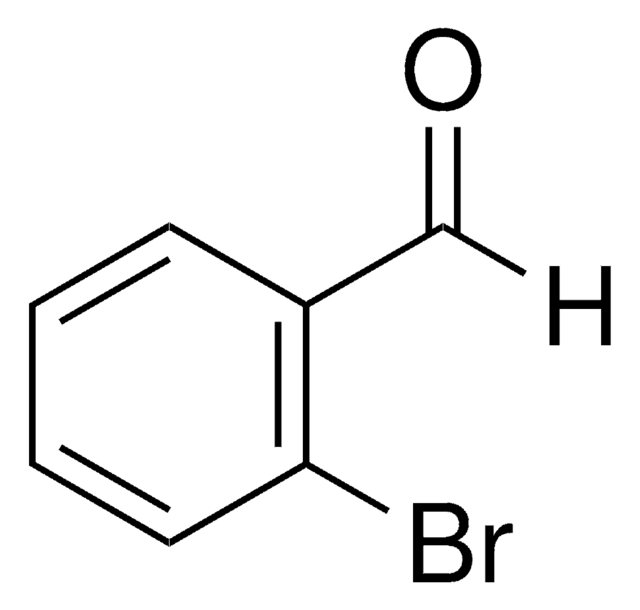
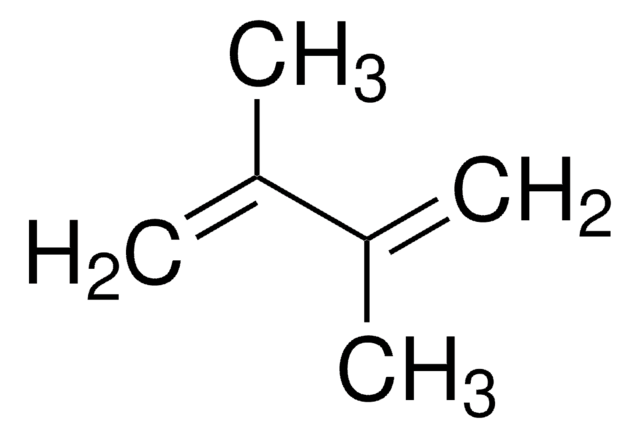
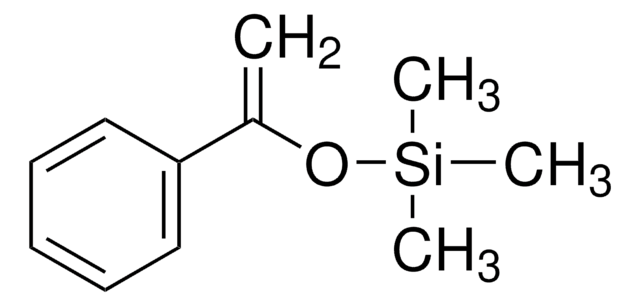
227226
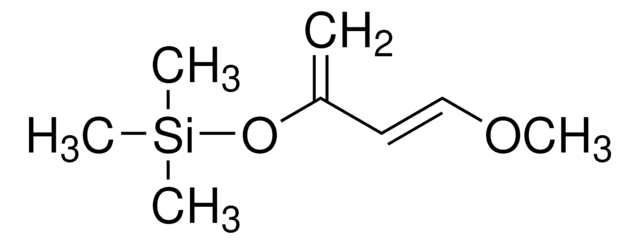
1-三甲基硅氧基-1,3-丁二烯
98%
别名:
(1,3-Butadien-1-yloxy)trimethylsilane, (1,3-Butadienyloxy)trimethylsilane, (Butadienyloxy)trimethylsilane, 1-(Trimethylsiloxy)butadiene, 1-[(Trimethylsilyl)oxy]-1,3-butadiene, 1-[(Trimethylsilyl)oxy]butadiene
About This Item
推荐产品
化驗
98%
形狀
liquid
折射率
n20/D 1.448 (lit.)
bp
131 °C (lit.)
密度
0.811 g/mL at 25 °C (lit.)
儲存溫度
2-8°C
SMILES 字串
C[Si](C)(C)O\C=C\C=C
InChI
1S/C7H14OSi/c1-5-6-7-8-9(2,3)4/h5-7H,1H2,2-4H3/b7-6+
InChI 密鑰
UQGOYQLRRBTVFM-VOTSOKGWSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
應用
訊號詞
Warning
危險分類
Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
3 - Flammable liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
77.0 °F - closed cup
閃點(°C)
25 °C - closed cup
個人防護裝備
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
其他客户在看
商品
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门