H5877
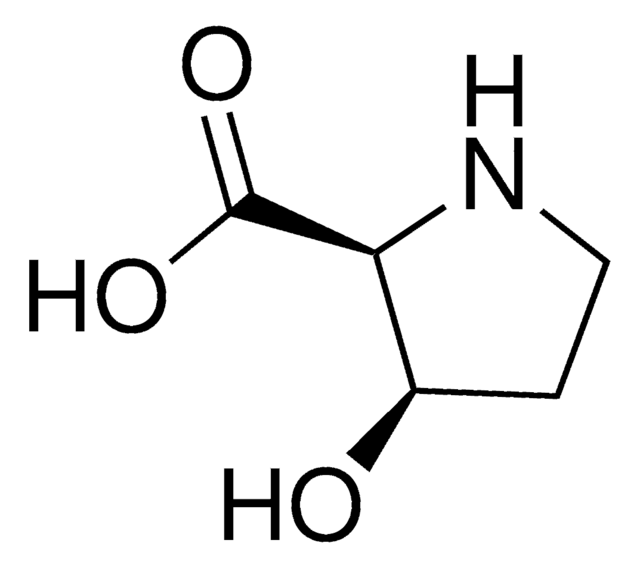
cis-4-Hydroxy-D-proline
≥98% (TLC), suitable for ligand binding assays
Synonym(s):
(2R,4R)-(+)-4-Hydroxy-2-pyrrolidinecarboxylic acid, D-allo-Hydroxyproline
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H9NO3
CAS Number:
Molecular Weight:
131.13
Beilstein:
81439
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Product Name
cis-4-Hydroxy-D-proline,
Assay
≥98% (TLC)
Quality Level
form
powder
technique(s)
ligand binding assay: suitable
color
white
mp
243 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
O[C@H]1CN[C@H](C1)C(O)=O
InChI
1S/C5H9NO3/c7-3-1-4(5(8)9)6-2-3/h3-4,6-7H,1-2H2,(H,8,9)/t3-,4-/m1/s1
InChI key
PMMYEEVYMWASQN-QWWZWVQMSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
Cis-4-Hydroxy-D-proline may be used as a starting material for the 13-step synthesis of new conformationally restricted PNA adenine monomer and the synthesis of N-Benzyl pyrrolidinyl sordaricin derivatives. Cis-4-Hydroxy-D-proline is a substrate that may be used to study the specificity and kinetics of D-alanine dehydrogenase. Cis-4-Hydroxy-D-proline may be used to analyze the substrate specificity of amino acid transporter PAT1.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A Püschl et al.
Organic letters, 2(26), 4161-4163 (2001-01-11)
[reaction:see text] A new conformationally restricted PNA adenine monomer has been synthesized in 13 steps from cis-4-hydroxy-D-proline. A fully modified adenine decamer displayed improved binding affinity toward complementary DNA and RNA oligonucleotides as compared to that of the parent PNA
Masami Arai et al.
Bioorganic & medicinal chemistry letters, 12(19), 2733-2736 (2002-09-10)
N-Benzyl pyrrolidinyl sordaricin derivatives have been synthesized from cis-4-hydroxy-D-proline in a stereocontrolled manner. These compounds maintained moderate antifungal activity against several pathogenic fungal strains. Their MIC values against Candida albicans were in the range of 0.25-2 microg/mL.
L Metzner et al.
Amino acids, 31(2), 111-117 (2006-05-16)
The proton coupled amino acid transporter PAT1 expressed in intestine, brain, and other organs accepts L- and D-proline, glycine, and L-alanine but also pharmaceutically active amino acid derivatives such as 3-amino-1-propanesulfonic acid, L-azetidine-2-carboxylic acid, and cis-4-hydroxy-D-proline as substrates. We systematically
Charles E Deutch
FEMS microbiology letters, 238(2), 383-389 (2004-09-11)
3,4-Dehydro-DL-proline is a toxic analogue of L-proline which has been useful in studying the uptake and metabolism of this key amino acid. When membrane fractions from Escherichia coli strain UMM5 (putA1::Tn5 proC24) lacking both L-proline dehydrogenase and L-Delta(1)-pyrroline-5-carboxylate reductase were
Ashley C Campbell et al.
Archives of biochemistry and biophysics, 698, 108727-108727 (2020-12-18)
Proline utilization A (PutA) proteins are bifunctional proline catabolic enzymes that catalyze the 4-electron oxidation of l-proline to l-glutamate using spatially-separated proline dehydrogenase and l-glutamate-γ-semialdehyde dehydrogenase (GSALDH, a.k.a. ALDH4A1) active sites. The observation that l-proline inhibits both the GSALDH activity
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service