F5126
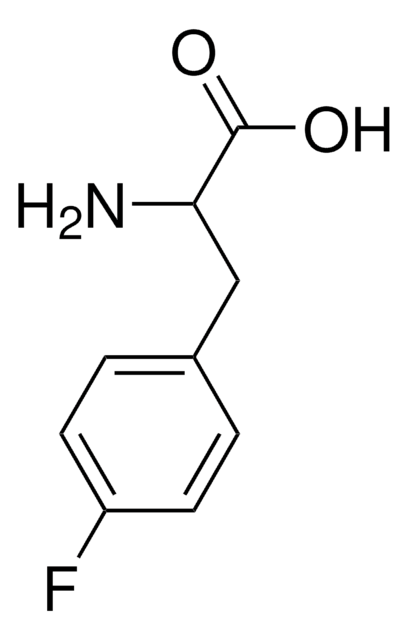
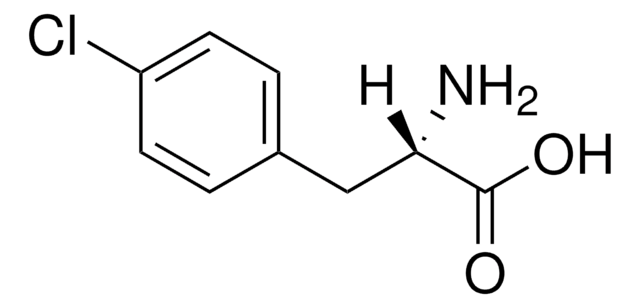
m-Fluoro-DL-phenylalanine
≥98%
Synonym(s):
3-Fluoro-DL-phenylalanine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
FC6H4CH2CH(NH2)COOH
CAS Number:
Molecular Weight:
183.18
Beilstein:
2939807
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Product Name
m-Fluoro-DL-phenylalanine,
Assay
≥98%
Quality Level
form
powder
color
white to off-white
mp
240-250 °C
application(s)
cell analysis
peptide synthesis
SMILES string
NC(Cc1cccc(F)c1)C(O)=O
InChI
1S/C9H10FNO2/c10-7-3-1-2-6(4-7)5-8(11)9(12)13/h1-4,8H,5,11H2,(H,12,13)
InChI key
VWHRYODZTDMVSS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
m-Fluoro-DL-phenylalanine, a toxic antimetabolite, is a racemic mixture of a substituted (halogenated) benzoyl D and L phenylalanine with potential use in antiviral and antimicrobial drug development.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Nick J P Wierckx et al.
Applied and environmental microbiology, 71(12), 8221-8227 (2005-12-08)
Efficient bioconversion of glucose to phenol via the central metabolite tyrosine was achieved in the solvent-tolerant strain Pseudomonas putida S12. The tpl gene from Pantoea agglomerans, encoding tyrosine phenol lyase, was introduced into P. putida S12 to enable phenol production.
Monica N Kinde et al.
Proceedings of the National Academy of Sciences of the United States of America, 113(48), 13762-13767 (2016-11-20)
Voltage-gated sodium channels (NaV) play an important role in general anesthesia. Electrophysiology measurements suggest that volatile anesthetics such as isoflurane inhibit NaV by stabilizing the inactivated state or altering the inactivation kinetics. Recent computational studies suggested the existence of multiple
Julianne L Kitevski-LeBlanc et al.
Journal of biomolecular NMR, 48(2), 113-121 (2010-08-25)
In protein NMR experiments which employ nonnative labeling, incomplete enrichment is often associated with inhomogeneous line broadening due to the presence of multiple labeled species. We investigate the merits of fractional enrichment strategies using a monofluorinated phenylalanine species, where resolution
Julianne L Kitevski-Leblanc et al.
Journal of biomolecular NMR, 47(2), 113-123 (2010-04-20)
Traditional single site replacement mutations (in this case, phenylalanine to tyrosine) were compared with methods which exclusively employ (15)N and (19)F-edited two- and three-dimensional NMR experiments for purposes of assigning (19)F NMR resonances from calmodulin (CaM), biosynthetically labeled with 3-fluorophenylalanine
H Ito et al.
Agricultural and biological chemistry, 54(3), 699-705 (1990-03-01)
A wild-type parent of Brevibacterium lactofermentum was converted into an L-Tyr producer by three steps of genetic breeding. First, acquirement of m-fluoro-D, L-phenylalanine resistance (1,000 microgram/ml) brought about MF1317 which produced 3.5 g/l of L-Tyr and a byproduct of 2.8
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service