C4805
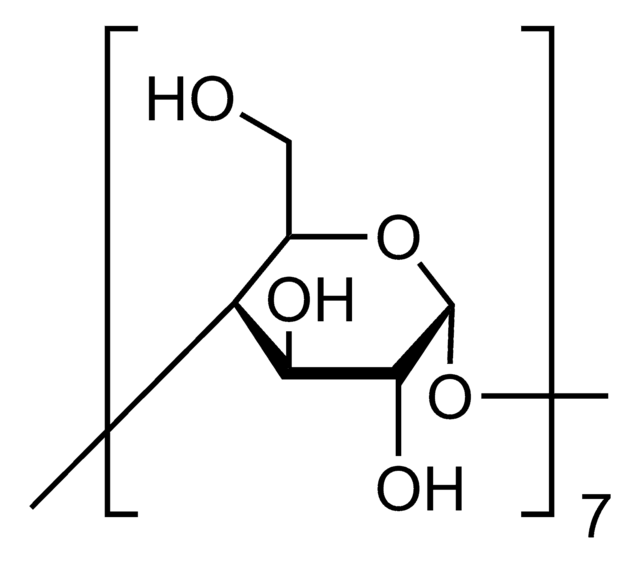
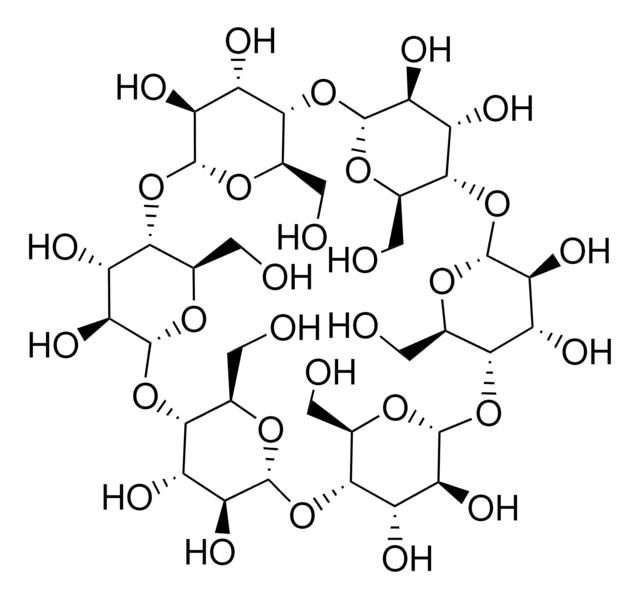
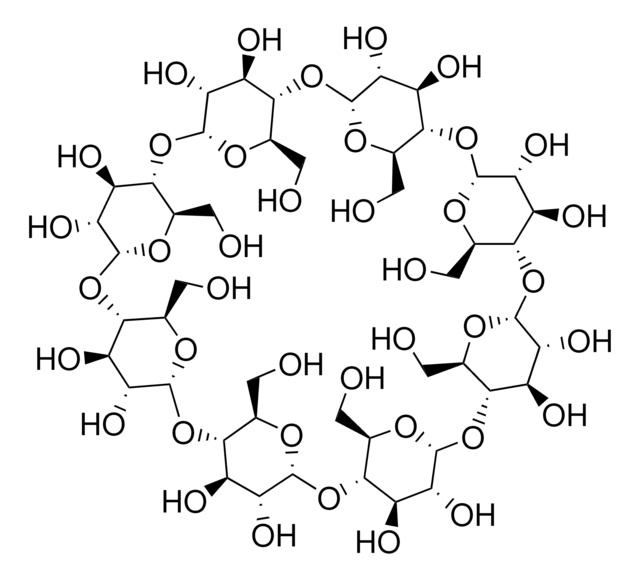
β-Cyclodextrin
powder, BioReagent, suitable for cell culture, ≥97%
Synonym(s):
Caraway, Cycloheptaamylose, Cyclomaltoheptaose, Schardinger β-Dextrin
About This Item
Recommended Products
sterility
non-sterile
product line
BioReagent
Assay
≥97%
form
powder
optical activity
[α]20/D +162±3°, c = 1.5% in H2O
technique(s)
cell culture | mammalian: suitable
mp
290-300 °C (dec.) (lit.)
solubility
1 M NaOH: 50 mg/mL
shipped in
ambient
storage temp.
room temp
SMILES string
OC[C@H]1O[C@@H]2O[C@H]3[C@H](O)[C@@H](O)[C@H](O[C@@H]3CO)O[C@H]4[C@H](O)[C@@H](O)[C@H](O[C@@H]4CO)O[C@H]5[C@H](O)[C@@H](O)[C@H](O[C@@H]5CO)O[C@H]6[C@H](O)[C@@H](O)[C@H](O[C@@H]6CO)O[C@H]7[C@H](O)[C@@H](O)[C@H](O[C@@H]7CO)O[C@H]8[C@H](O)[C@@H](O)[C@H](O[C@@H]8CO)O[C@H]1[C@H](O)[C@H]2O
InChI
1S/C42H70O35/c43-1-8-29-15(50)22(57)36(64-8)72-30-9(2-44)66-38(24(59)17(30)52)74-32-11(4-46)68-40(26(61)19(32)54)76-34-13(6-48)70-42(28(63)21(34)56)77-35-14(7-49)69-41(27(62)20(35)55)75-33-12(5-47)67-39(25(60)18(33)53)73-31-10(3-45)65-37(71-29)23(58)16(31)51/h8-63H,1-7H2/t8-,9-,10-,11-,12-,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23-,24-,25-,26-,27-,28-,29-,30-,31-,32-,33-,34-,35-,36-,37-,38-,39-,40-,41-,42-/m1/s1
InChI key
WHGYBXFWUBPSRW-FOUAGVGXSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
How palmitic acid, other fatty acids and other cell culture components affect the performance of serum-free, protein-free cell culture systems used for biomanufacturing heterologous proteins including monoclonal antibodies.
How the unsaturated fatty acid, oleic acid and other cell culture components affect the performance of serum-free, protein-free cell culture systems used for biomanufacturing heterologous proteins including monoclonal antibodies.
Many metabolically important compounds, such as lipid-soluble vitamins and hormones, have very low solubilities in aqueous solutions. Various techniques have been used to solubilize these compounds in tissue culture, cell culture, or other water-based applications.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service