T81604
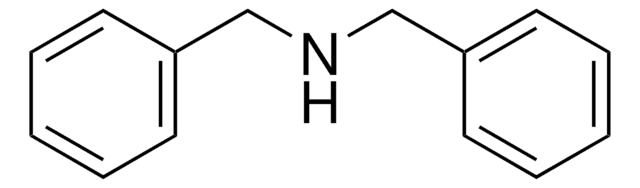
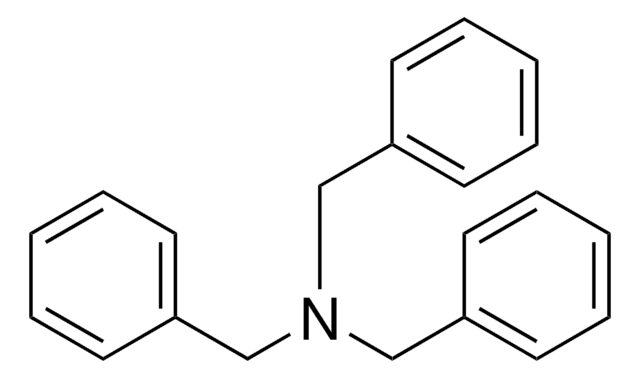
Triphenylamine
98%
Synonym(s):
N,N-Diphenylaniline, N,N-Diphenylbenzenamine
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(C6H5)3N
CAS Number:
Molecular Weight:
245.32
Beilstein:
2050487
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
bp
347-348 °C (lit.)
mp
124-128 °C (lit.)
SMILES string
c1ccc(cc1)N(c2ccccc2)c3ccccc3
InChI
1S/C18H15N/c1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18/h1-15H
InChI key
ODHXBMXNKOYIBV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Photoinitiators for Hydrogel Formation: Triphenylamine derivatives are used as novel D-pi-A hemicyanine dyes, acting as effective photoinitiators for in situ hydrogel formation and DLP printing, expanding their applications in biomedical engineering and 3D printing technologies (Du et al., 2024).
- Supramolecular Photosensitizers: A supramolecular construct based on triphenylamine and pyrazine demonstrates aggregation-induced emission properties, enhancing the efficiency of photooxidation reactions. This development offers potential improvements in photodynamic therapy and environmental applications (Dong et al., 2024).
- Highly Efficient OLEDs: Triphenylamine is integral in synthesizing new phenanthro[9,10-d]oxazole-based fluorophores with hybridized local and charge-transfer characteristics. These materials are crucial for developing blue non-doped OLEDs with minimal efficiency roll-off, significant for advanced display technologies (Xie et al., 2024).
- Memory Device Applications: Modifications in donor end caps in N-heteroaromatic systems containing triphenylamine were explored for binary-to-ternary WORM memory conversion, contributing to advancements in memory storage technology (Gayathri et al., 2024).
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Jiu-Qiang Huang et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 154, 164-170 (2015-11-02)
A new series of trans-alkenes (3a-3e) containing different electron-donating groups were synthesized by the solvent-free Horner-Wadsworth-Emmons reaction, and characterized by infrared, hydrogen nuclear magnetic resonance, mass spectrometry and elemental analysis. Their UV-visible absorption, one-photon excited fluorescence, two-photon absorption, and two-photon
Minggang Tian et al.
Organic & biomolecular chemistry, 12(32), 6128-6133 (2014-07-06)
Cysteine (Cys) plays important roles in many physiological processes of eukaryotic cells and its detection in cells is of fundamental significance. However, glutathione (GSH), homocysteine, N-acetyl-L-cysteine and other thiols greatly hamper the detection of Cys. In particular, GSH strongly interferes
Xu Jing et al.
Journal of the American Chemical Society, 137(11), 3967-3974 (2015-03-05)
The design of artificial systems that mimic highly evolved and finely tuned natural photosynthetic systems is a subject of intensive research. We report herein a new approach to constructing supramolecular systems for the photocatalytic generation of hydrogen from water by
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service