291110
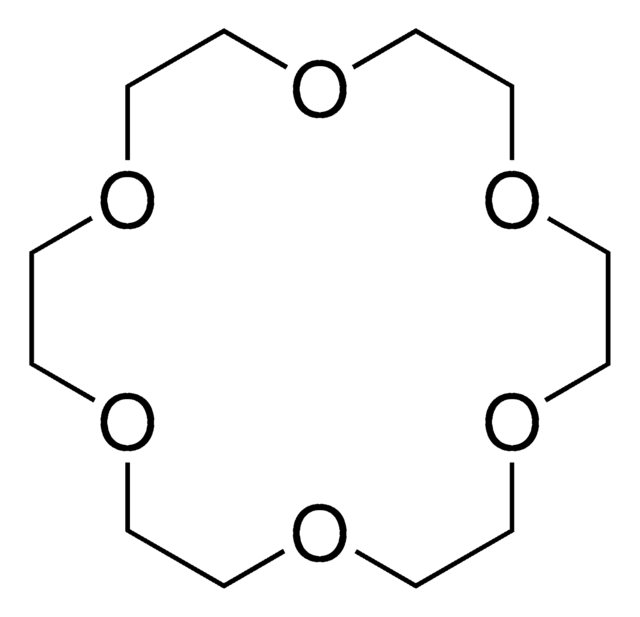
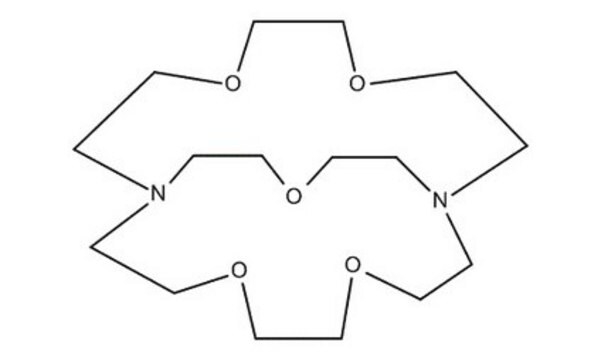
4,7,13,16,21,24-Hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane
98%
Synonym(s):
Cryptand 222, Kryptofix® 222
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C18H36N2O6
CAS Number:
Molecular Weight:
376.49
Beilstein:
620282
EC Number:
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
mp
68-71 °C (lit.)
SMILES string
C1COCCN2CCOCCOCCN(CCO1)CCOCCOCC2
InChI
1S/C18H36N2O6/c1-7-21-13-14-24-10-4-20-5-11-25-17-15-22-8-2-19(1)3-9-23-16-18-26-12-6-20/h1-18H2
InChI key
AUFVJZSDSXXFOI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
4,7,13,16,21,24-Hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane(cryptand 222, Kryptofix 222) is an organic compound with formula C18H36N2O6. This compound has a cage-like three-dimensional structure that donates N2O6. The ligand has shown a wide range of uses in magnetic resonance imaging, organic synthesis, crystallography, electrochemistry, and chromatography. In solution, it is a well-known sequestering agent for metal ions.
Application
4,7,13,16,21,24-Hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane is used as:
- A electrolyte additive in isotachophoresis[separation method that is particularly suited to the analysis of small ions]for the analysis of alkali metal cations.
- A complexing agent in the preparation of K+-imprinted nanoparticles using methacrylic acid as the functional monomer, ethylene glycol dimethacrylate as the crosslinker and 2,2′-azobisisobutyronitrile as the radical initiator.
- A structure directing agent (SDA) in the preparation of LTA-type AlPO4 crystals.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Cryptand used with potassium mirror to reduce a hindered distannene to a crystalline radical anion.
Legal Information
Kryptofix is a registered trademark of Merck KGaA, Darmstadt, Germany
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Metal Ion Compiexing by Cryptand 222 in Solutions. A Thermodynamic Approach
Marcus Y
Reviews in Analytical Chemistry, 23, 269-302 (2004)
Preparation of large and well-shaped LTA-type AlPO4 crystals by using crown ether Kryptofix 222 as structure directing agent
Huang A, et al.
Microporous and Mesoporous Materials : The Official Journal of the International Zeolite Association, 129, 90-99 (2010)
Vladimir Ya Lee et al.
Journal of the American Chemical Society, 128(35), 11643-11651 (2006-08-31)
((t)Bu(2)MeSi)(2)Sn=Sn(SiMe(t)Bu(2))(2) 1, prepared by the reaction of (t)Bu(2)MeSiNa with SnCl(2)-diox in THF and isolated as dark-green crystals, represents the first example of acyclic distannene with a Sn=Sn double bond that is stable both in the crystalline form and in solution.
T Colomina et al.
Veterinary and human toxicology, 33(2), 121-124 (1991-04-01)
The effects of repeated ip administration of diethylenetriaminepentaacetic acid (DTPA), Kryptofix 222, 1,4,7,10,13,16-hexaoxacyclooctadecane (18-crown-6), ethylenglycol-bis-(beta-amino-ethylether)-N,N'-tetraacetic acid (EGTA), and Kryptofix 5 on the distribution and excretion of sc-injected strontium were investigated in male Swiss mice. Groups of 20 animals received 95
T Chaly et al.
International journal of radiation applications and instrumentation. Part B, Nuclear medicine and biology, 16(4), 385-387 (1989-01-01)
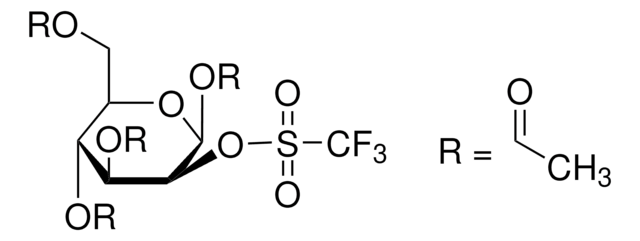
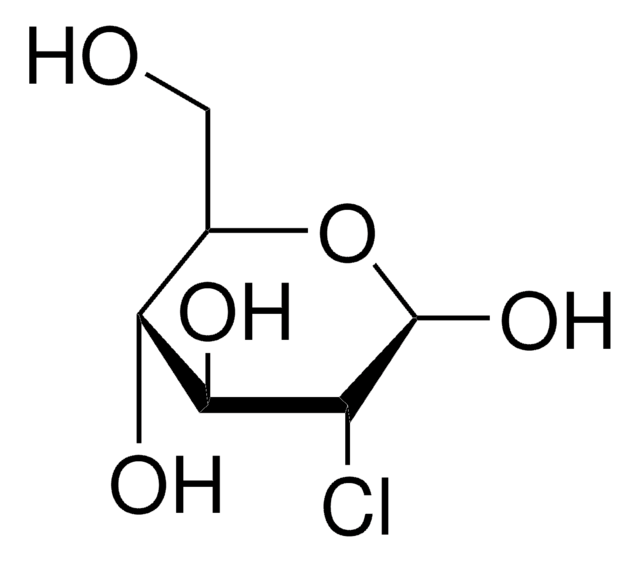
A simple procedure for the detection of 4,7,13,16,21,24-hexaoxa-1,10-diazabicyclo (8.8.8) hexacosane (Kryptofix 2.2.2) in the final solution of [18F]FDG prepared by the aminopolyether supported nucleophilic substitution of 1,3,4,6-tetra-O-acetyl 2-O-trifluoromethanesulfonyl-beta-D-mannopyranose (Hamacher et al., 1986) has been developed. Presence of Kryptofix 2.2.2 in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service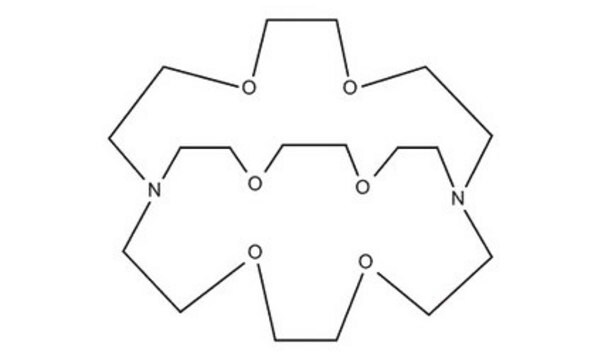

![4,7,13,16,21-Pentaoxa-1,10-diazabicyclo[8.8.5]tricosane 98%](/deepweb/assets/sigmaaldrich/product/structures/444/464/eeb08f63-862e-447e-8e41-342d713f439c/640/eeb08f63-862e-447e-8e41-342d713f439c.png)